Advances in Animal and Veterinary Sciences
Research Article
The Antinociceptive and Antipyretic Potentials of Cefepime
Mohamed Elbadawy1*, Hussein M. El-Husseiny2, Mossad Gamaleddin Ahmed Elsayed1, Ashraf A. Elkomy1, Amira Abugomaa3*
1Department of Pharmacology, Faculty of Veterinary Medicine, Benha University, 13736 Moshtohor, Toukh, Elqaliobiya, Egypt; 2Department of Surgery, Anesthesiology, and Radiology, Faculty of Veterinary Medicine, Benha University, Moshtohor, Toukh 13736, Elqaliobiya, Egypt; 3Faculty of Veterinary Medicine, Mansoura University, 35516 Mansoura, Dakahliya, Egypt.
Abstract | The present study aimed to evaluate the analgesic and antipyretic potentials of cefepime at different therapeutic doses in vivo using hot-plate and brewer’s yeast-induced fever models in mice and rats, respectively. Twenty-five mice and/or rats were assigned into five groups, each of five animals. The first group of each was left as control and administered saline i.m. The second group was given ketoprofen (13 mg.) or metamizole sodium (150 mg) per kg BW i.m. as a standard analgesic and antipyretic, respectively. The third, fourth, and fifth groups were given cefepime at the different therapeutic doses, respectively, i.m. in saline. Cefepime induced dose-dependent and significant analgesic effect along the four h of the experiment which was indicated by the longer reaction time in treated than the control group. Cefepime at 45 mg/kg. BW had no antipyretic activity, while at 90 and 180 mg/kg BW had an antipyretic effect after 3.5 h of cefepime administration and at 180 mg/kg BW had a significant antipyretic potential 2.5 h after its injection. These findings indicate that cefepime possesses significant analgesic and antipyretic potentials that would be valuable in symptomatic relief of nociception and fever besides its well-established antibacterial effect.
Keywords | Analgesic, Antinociceptive, Antipyretic, Cefepime, Cephalosporins.
Received | August 29, 2021; Accepted | September 05, 2021; Published | November 01, 2021
*Correspondence | Mohamed Elbadawy, Amira Abugomaa, Department of Pharmacology, Faculty of Veterinary Medicine, Benha University, 13736 Moshtohor, Toukh, Elqaliobiya, Egypt; Faculty of Veterinary Medicine, Mansoura University, 35516 Mansoura, Dakahliya, Egypt; Email: mohamed.elbadawy@fvtm.bu.edu.eg; s193249s@st.go.tuat.ac.jp
Citation | Elbadawy M, El-Husseiny HM, Elsayed MGA, Elkomy AA, Abugomaa A (2021). The antinociceptive and antipyretic potentials of cefepime. Adv. Anim. Vet. Sci. 9(12): 2132-2138.
DOI | http://dx.doi.org/10.17582/journal.aavs/2021/9.12.2132.2138
ISSN (Online) | 2307-8316; ISSN (Print) | 2309-3331
Copyright © 2021 Elbadawy et al. This is an open access article distributed under the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
INTRODUCTION
Pain and fever are major symptoms of many illnesses caused by intense or damaging stimuli (Raja et al., 2020) or microbial infection (Bernheim et al., 1979). Nociception is the reaction of sensory nerve endings to such stimuli whose intensity is close to or surpassing noxious intensity (nociceptors). Damages to the peripheral nerve endings, spinal cord, or brain can arouse different responses resulting in the behaviour of pain (Merskey and Bogduk, 1994). Fever evolves in patients upon certain exogenous stimuli (mostly microbial) that stimulate bone-marrow-derived phagocytes to discharge fever-inducing hormones (endogenous pyrogens) as a mechanism to control infection (Bernheim et al., 1979). An analgesic is a drug that relieves symptoms of pain (Tambaro et al., 2013; Tripathi, 2013). Analgesic drugs act either peripherally or centrally or on both. Analgesics include non-steroidal anti-inflammatory agents (NSAIDs) such as aspirin, and opioid agents such as codeine, morphine, and many others. Antipyretics are drugs that are administered to treat fever by triggering the hypothalamus to reverse the prostaglandin-caused rise in body temperature (BT). The selection of each depends on the severity and nature of pain, patient status, and co-administered drugs (WHO, 2006).
Other than the available standard analgesics and antipyretics, many other agents may have analgesic and antipyretic potentials or other along with their basic pharmacological effect (Aboubakr et al., 2014). For example, some antibiotics, regardless its type, were demonstrated to kill pain to various levels in rats (Elbadawy, 2007; Ocana and Baeyens, 1991; Suaudeau et al., 1993). This highlights the advantage of synergism when these antibiotics are co-given with the standard analgesics and antipyretics, making sometimes, therapy more effective.
Bacterial infections can change the quality or even threaten the lives of human beings and livestock or result in serious economic losses; therefore, antibacterial intervention is recommended (Elbadawy et al., 2019). Antibiotics are major drugs in many prescriptions of bacterial infection. Antibiotics have many side effects, which are sometimes harmful (Aboubakr et al., 2014; El-Safty et al., 2018; Elkomy et al., 2020; Elsayed et al., 2014) or beneficial to some patients (Aboubakr and Elbadawy, 2016; Aboubakr et al., 2014; El-Mahmoudy and Gheith, 2016; Elbadawy and Aboubakr, 2017; Elbadawy et al., 2021; Elkomy et al., 2019; Elsayed et al., 2014). Cefepime, a parenteral 4th-generation cephalosporin, is a well-established, generally well-tolerated antibiotic (Okamoto et al., 1993), with broad-spectrum activity and less bacterial resistance (Yayan et al., 2016). Cefepime is active in vitro against diverse bacteria and is stable against numerous beta-lactamases (Nguyen et al., 2014). Cefepime was reported to have a low potential to produce adverse reactions at therapeutic doses (Elsayed et al., 2013; Elsayed et al., 2014).
Expanded information concerning the beneficial side actions of cefepime will be of benefit to physicians and patients. Therefore, the present study aimed to assess the analgesic and antipyretic potentials of cefepime in vivo.
MATERIALS AND METHODS
Drug (cefepime)
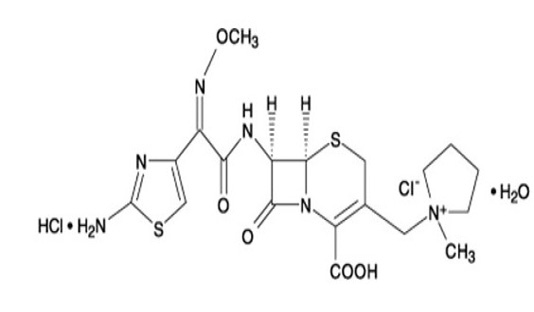
cefepime is a semi-synthetic, broad-spectrum, 4th-generation cephalosporin antibiotic formulated for injection. The chemical formula is 1-[[(6R,7R)-7-[2-(2-amino-4-thiazolyl)-glyoxylamido]-2-carboxy-8-oxo-5-thia-1-azabicyclo[4.2.0]oct-2-en-3-yl]methyl]-1-methylpyrrolidinium chloride, 72-(Z)-(O-methyloxime), monohydrochloride, monohydrate, which corresponds to structure shown at Figure 1.
Cefepime hydrochloride is a white to pale yellow powder with a molecular formula of C19 H25 Cl N6 O5 S2·HCl·H20 and a molecular weight of 571.5. It is highly soluble in water. Cefepime hydrochloride is supplied for i.m. or i.v. injection in a strength equivalent to 500, 1000 m, or 2000 mg of cefepime. It is produced by GSK group Co., Cairo, Egypt with the commercial name Maxipime®.
Animals
Experiments were done on mice and rats for assessing the analgesic and antipyretic potentials of cefepime. Animals were purchased from the Experimental Animal Laboratory of the Faculty of Veterinary Medicine, Benha University, Egypt. Animals were maintained in polypropylene cages with wood bedding in a controlled room with 24°C and relative humidity of 60% and a 12h light/dark cycle. Standard pellet diet and water were supplied ad libitum. Animals were kept for one week to adapt to the environment before experimentation. The experimental steps were performed following the guidelines set by the local Ethical Committee of Faculty of Veterinary Medicine, Benha University, Egypt. All efforts were followed to maintain the rest of the animals.
Experimental design
In the present investigation, mice and rats were used to assess the analgesic and antipyretic potentials of cefepime, respectively. For assessing the analgesic potential of cefepime, Numerous Swiss albino mice were tested for the ordinary response to thermal stimuli-provoked pain, and only ordinarily reacting ones were selected for the pain experiments. Twenty-five mice of both sexes and 20-25 g BW were used and distributed into five groups, each of five mice. The first one was left as a control and was given normal saline i.m. The second group was given ketoprofen (Orudis®) as a standard analgesic at 13 mg/kg BW i.m. in saline. The third, fourth, and fifth groups were administered cefepime at 65, 130, and 260 mg/kg BW i.m. in saline as small, medium, and large therapeutic doses, respectively. The doses were calculated following the Paget and Barnes table of dose conversion from humans to mice (Paget and Barnes,
Table 1: Effects of cefepime (65, 130, and 260 mg/kg BW i.m.) and ketoprofen (13 mg/kg BW i.m.) on the reaction time (seconds) induced in the hot-plate test in mice (mean ± S.E.; n = 5).
| Groups | Before experiment | Time post-injection (h) | ||||||||
| 0.5 | 1 | 1.5 | 2 | 2.5 | 3 | 3.5 | 4 | |||
Reaction time (seconds) | ||||||||||
| Control group | 7.18 ± 0.27 | 6.29 ± 0.24 | 6.80 ± 0.22 | 6.72 ± 0.40 | 6.46 ± 0.36 | 5.85 ± 0.53 | 6.52 ± 0.41 | 6.58 ± 0.48 | 6.82 ± 0.74 | |
Ketoprofen (13 mg/kg BW) | 6.49 ± 0.17 | 17.7 ± 0.45 *** ΔΔΔ | 16.8 ± 0.97 *** ΔΔΔ | 16.2± 0.58 *** ΔΔΔ | 16.4 ± 1.03 *** ΔΔΔ | 13.5 ± 1.3 *** ΔΔΔ | 13.5 ± 1.32 *** ΔΔΔ | 13.7 ± 0.69 *** ΔΔΔ | 11.8 ± 0.34 *** ΔΔΔ | |
Cefepime (65 mg/kg BW) | 6.13 ± 0.28 | 8.61 ± 0.58 ** ΔΔ | 9.75 ± 0.39 *** ΔΔΔ | 10.1 ± 0.32 *** ΔΔΔ | 10.2 ± 0.15 *** ΔΔΔ | 10.3 ± 0.35 *** ΔΔΔ | 10.2 ± 0.50 *** ΔΔΔ | 10.6 ± 0.41 ** ΔΔΔ | 11.4 ± 0.30 *** ΔΔΔ | |
Cefepime (130 mg/kg BW) | 6.44 ± 0.55 | 8.99 ± 0.30 *** ΔΔ | 10.8 ±0.51 *** ΔΔΔ | 11.1 ± 0.28 *** ΔΔΔ | 12.1 ± 0.26 *** ΔΔΔ | 13.0 ± 0.53 *** ΔΔΔ | 12.8 ±1.05 *** ΔΔΔ | 13.1 ± 0.59 *** ΔΔΔ | 13.2 ± 1.06 ** ΔΔΔ | |
Cefepime (260 mg/kg BW) | 6.66 ± 0.28 | 9.46 ± 0.30 *** ΔΔΔ | 10.2 ± 0.25 *** ΔΔΔ | 10.7 ± 0.35 *** ΔΔΔ | 11.0 ± 0.25 *** ΔΔΔ | 12.6 ± 0.44 *** ΔΔΔ | 11.8 ± 0.52 *** ΔΔΔ | 12.3 ± 0.18 *** ΔΔΔ | 13.6 ± 0.64 *** ΔΔΔ | |
* → Represents the significance with the data of the control group.
Δ → Represents the significance in comparison with data of the same group before treatment.
* or Δ P> 0.05 ** or ΔΔ P> 0.01 *** or ΔΔΔ P> 0.001
Table 2: Effect of cefepime (45, 90, and 180 mg/kg BW i.m.) and metamizole sodium (150 m/kg BW i.m.) on body temperature (°C) after Brewer’s yeast- induced pyrexia in rats (mean ± S.E.; n = 5).
Groups | Before experiment | After Brewer’s yeast | Time after injection (h) | |||||||
| 0.5 | 1 | 1.5 | 2 | 2.5 | 3 | 3.5 | 4 | |||
Rectal temperature (°C) | ||||||||||
| Control group | 36.7 ± 0.1 | 37.7 ± 0.04 | 37.5 ± 0.12 | 37.4 ± 0.12 | 37.5 ± 0.13 | 37.4 ± 0.13 | 37.6 ± 0.12 | 37.5 ± 0.12 | 37.7 ± 0.11 | 37.4 ± 0.13 |
Metamizole (150 mg/kg BW) | 36.4 ± 0.09 | 37.7 ± 0.09 | 37.1 ± 0.13 ΔΔ | 36.7 ± 0.10 ** ΔΔΔ | 36.6 ± 0.1 *** ΔΔΔ | 36.5 ± 0.09 *** ΔΔΔ | 36.4 ± 0.07 *** ΔΔΔ | 36.4 ± 0.09 *** ΔΔΔ | 36.4 ± 0.09 *** ΔΔΔ | 36.4 ± 0.09 *** ΔΔΔ |
Cefepime (45 mg/kg BW) | 36.6 ± 0.1 | 37.7 ± 0.1 | 37.6 ± 0.07 | 37.5 ± 0.08 | 37.5 ± 0.06 | 37.5 ± 0.06 | 37.4 ± 0.07 Δ | 37.3 ± 0.07 Δ | 37.3 ± 0.09 Δ | 37.2 ± 0.09 Δ |
Cefepime (90 mg/kg BW) | 36.4 ± 0.05 | 37.6 ± 0.05 | 37.5 ± 0.05 | 37.5 ± 0.05 | 37.4 ± 0.05 Δ | 37.4 ± 0.05 ΔΔ | 37.3 ± 0.05 ΔΔΔ | 37.2 ± 0.03 ΔΔΔ | 37.1 ± 0.04 * ΔΔΔ | 37.0 ± 0.02 * ΔΔΔ |
Cefepime (180 mg/kg BW) | 36.5 ± 0.07 | 37.6 ± 0.07 | 37.5 ± 0.04 | 37.4 ± 0.02 Δ | 37.4 ± 0.02 ΔΔ | 37.3 ± 0.04 ΔΔ | 37.1 ± 0.04 * ΔΔΔ | 37.0 ± 0.05 ** ΔΔΔ | 36.9 ± 0.04 ** ΔΔΔ | 36.8 ± 0.06 ** ΔΔΔ |
* → Represents the significance with the data of the control group.
* or Δ P> 0.05 ** or ΔΔ P> 0.01 *** or ΔΔΔ P> 0.001
1964). The hot-plate test was then performed to check the analgesic potential of cefepime.
For assessing the antipyretic potential of cefepime, 25 Albino rats of both sexes weighing 150-200 g BW were used. They were distributed into five groups, each of five rats. All rats were made hyperthermic by s.c. injection of 20 % suspension of Brewer’s yeast in physiological saline at 0.1 ml /100 g BW. The method described before (Alperman, 1972; Brune and Alpermann, 1983) was followed for studying the impact of cefepime on feverish BT.
Hot-Plate Test
The analgesic potential of cefepime was assessed using the hot-plate method as demonstrated before (Eddy et al., 1950) (Elbadawy et al., 2021). After treatment of mice as mentioned above, each mouse was placed singly in a 2-liter volume beaker on a hot plate (Scilogex, CT, U.S.A) kept constant at 55°C for determining the analgesic potential of cefepime. The time elapsed (in seconds) until the mouse jumps or licks its paws (as responses to the thermal stimulus-triggered pain sensation) was recorded at 1, 2, 3, and 4 h post cefepime administration and considered as the reaction time. A “cutoff” time of 30 seconds was followed to prevent tissue harm to mice.
Antipyretic potential of cefepime
Seventeen h post yeast injection, the initial BT of each rat was recorded rectally using a medical thermometer. Normal saline was given i.m to the first group and was kept as control. The second group was given metamizole sodium (Novalgin®) as a standard antipyretic drug at 150 m/kg BW i.m. as a standard antipyretic. The third, fourth, and fifth groups were administered cefepime at a dose of 45, 90, and180 mg/kg BW i.m. in saline as small, medium, and large therapeutic doses, respectively, based on Paget and Barnes dose conversion from human to rats (Paget and Barnes, 1964). The BT of each rat was then recorded before and at 30, 60, 90, 120, 150, 180, 210, 240 min after administrations. The difference in BT between the treated and control groups was taken as a measure of antipyretic activity.
Statistical Analysis
Data were expressed as mean ± S.E. The obtained data were analyzed by the Student t-test to express the differences between groups (Snedecor and Cochran, 1980). Each treated group was compared with the control one and itself before treatment. Comparison of the mean values was carried out and differences were considered statistically significant when P<0.05.
RESULTS
Analgesic potential of cefepime in mice
The analgesic-like action of cefepime in mice is recorded in the Table (1). Intramuscular injection of cefepime in all tested doses caused a significant analgesic effect along the four h of the experiment which was indicated by the longer reaction time in treated groups compared with the control group and same before treatment.
Antipyretic activity of cefepime in rats
Seventeen h after injection of Brewers yeast, all rats showed hyperthermia (Table 2). The antipyretic-like action of cefepime in rats is recorded in Table 2. Intramuscular injection of cefepime at 45 mg/kg BW had no antipyretic activity while at 90 mg/kg BW had an antipyretic effect started at 3.5 h of cefepime administration. The cefepime in a dose of 180 mg/kg BW had a significant antipyretic potential started at 2.5 h of injection.
DISCUSSION
Analgesics, antipyretics, and others are generally prescribed along with antibiotics for the remediation of systemic infections with fever and pain. However, many antibiotics including cefepime have other pharmacological actions like reducing spontaneous locomotor activity, relaxing the skeletal muscles, stimulating intestinal movements, potentiating anesthesia in mice, inducing significant hypothermia and analgesia (Elsayed et al., 2013; Goto et al., 1992; Kadota et al., 1992). Hasegawa et al. showed that cefadroxil inhibited gastric secretion and raised the biliary secretion in rats (Hasegawa et al., 1979). Takai et al. also showed that cefoperazone has several side actions at different doses in different laboratory animals (Takai et al., 1980). Cefamandole caused very marked hypotensive effects and a decline in respiration rate in anesthetized dogs (El-Sayed et al., 1997). Thus investigating the other actions of drugs is important to predict whether it has favorable or unfavorable side effects. The administration of such antibiotics with favorable side actions may add synergism with the co-prescribed drugs and fasten the cure of the patients.
The present investigation was performed to assess analgesic and antipyretic potentials of cefepime. Cefepime at 65, 130, and 260 mg/kg BW induced a significant analgesic effect against thermal stimuli. This was indicated by a longer reaction time in the hot-plate test. The obtained result was consistent with that reported by Goto et al. (Goto et al., 1992) who observed that significant analgesia was noticed at the highest dose of cefepime in mice. Similarly, some beta-lactams demonstrated various analgesia degrees with various intensity of actions based on their administered amounts (Suaudeau et al., 1993). In animals, frequent giving of ceftriaxone was shown to diminish visceral and neuropathic pain (Gunduz et al., 2011; Hu et al., 2010; Lin et al., 2011; Yang et al., 2011) and lower the tolerance to the analgesic effect of nicotine (Schroeder et al., 2011) or morphine (Rawls et al., 2010) by upregulating glutamate transporter-1 (GLT-1) expression. Macaluso et al. revealed that a single intraperitoneal (i.p.) giving of ceftriaxone at 200 mg/kg BW induced analgesia in mouse models of postsurgical or inflammatory pain, via upregulating GLT-1 level in the spinal cord (Macaluso et al., 2013). They also demonstarted that ceftriaxone-produced nociception was additive to that induced by blocking of metabotropic glutamate5 receptors, which are stimulated by the extrasynaptic glutamate. Further, single i.v. injection of ceftriaxone (2000 mg) in human patients undergoing surgery for decompression of nerves induced analgesia revealed by a 10-fold substantial increase in the pain threshold for 4-6 h after surgery, whereas cefazoline did not (Macaluso et al., 2013). The GLT-1 regulates glutamate homeostasis which is included in the establishment and progress of pathological pain (Hu et al., 2010). The chronic constriction injury of the sciatic nerve lowered the level of GLT-1 in the dorsal horns of the spinal cord leading to significant hyperalgesia which was reversed by the i.p. injection of ceftriaxone (Hu et al., 2010). Further, they found that the intrathecal injection of ceftriaxone led to the specific GLT-1 overexpression and glutamate uptake in the spinal dorsal and similar antinociceptive effects to those of i.p. injection of ceftriaxone (Hu et al., 2010). In another study, cefepime was reported to interfere with metabotropic glutamate receptor pathways and increase glutamate leading to analgesia induction (Han et al., 2018). Therefore the analgesia induced by cefepime in the current study might be due to upregulation of spinal GLT-1 expression and its function.
In the current study, cefepime at 90 and 180 mg/kg BW showed significant antipyretic potential. This was evidenced by the decline in rat’s feverish BT induced after s.c. administration of Brewer’s yeast suspension. This effect seemed to be mediated centrally through an action on the heat-regulating center in such a manner to increase heat loss by peripheral vasodilatation of skin blood vessels, as well as the direct vascular relaxant and negative inotropic effect of cefepime (Elsayed et al., 2013). This finding is consistent with that recorded by Goto et al who recorded cefepime-induced significant hypothermia in mice (Goto et al., 1992). The obtained data was inconsistent with that of Takai et al. (1982) and Honda et al. (1980). They reported that cefbuperazone and ceftizoxime, respectively, did not affect BT in rabbits and mice (Honda et al., 1980; Takai et al., 1982). Also, Takai et al. stated that cefoperazone caused pyrexia in rabbits at 1,000 mg/kg (Takai et al., 1980). Hirai et al. (1986) also confirmed that cefteram induced only a slight elevation of BT in rabbits (Hirai et al., 1986).
CONCLUSION
The data obtained in the current study indicates that cefepime might have the potentials of being an analgesic and antipyretic agent along with its antibacterial action. However, the detailed mechanism of action is a future perspective. Thus, the administration of cefepime may support the actions of the co-prescribed standard analgesics and antipyretics. Furthermore, the reported findings explain the eminent effectiveness of cefepime in bacterial infections which are frequently associated with pain and/or pyrexia.
AUTHORS CONTRIBUTION
Mohamed Elbadawy: designed the study, methodology, and analyzed the data. Hussein M. El-Husseiny: methodology. Mossad Gamaleddin Ahmed Elsayed: study design and supervision. Ashraf A. Elkomy: revised the manuscript. Amira Abugomaa: methodology, analysis, and writing the draft of the manuscript. All authors wrote, revised, and approved the manuscript.
ACKNOWLEDGMENTS
None to declare.
CONFLICT OF INTEREST
The authors declared no potential conflicts of interest.
References