Effect of Different Diet Concentrations on Longevity and Fecundity of Parasitic Wasp Bracon hebetor (Say.) (Hymenoptera: Braconidae)
Effect of Different Diet Concentrations on Longevity and Fecundity of Parasitic Wasp Bracon hebetor (Say.) (Hymenoptera: Braconidae)
Sumra Ashraf1, Zain ul Abdin1,*, Saqi Kosar Abbas2, Rao Sohail Ahmad Khan3, Muhammad Tahir1, Sehrish Rasool1, Maryam Anwar1 and Fiaz Hussain1
1Department of Entomology, University of Agriculture, Faisalabad, 38040, Pakistan
2Bahauddin Zakariya University, Bahadur Sub-Campus, Layyah, Pakistan
3Centre of Agricultural Biochemistry and Biotechnology, University of Agriculture, Faisalabad, 38040, Pakistan
ABSTRACT
To enhance their life period and reproductive potential, the adults of parasitoids mostly depend on supplemental food sources, such as sugars and other carbohydrates. These food resources are commonly obtained from animal secretions or plant exudates which include honeydew, fruit juices and both floral and extra-floral nectar. A direct behavioral assay was conducted to investigate the dietary preference and effects of different diets on the fecundity, sex ratio and longevity of B. hebetor. Three different diets [Honey syrup, sugar syrup, date syrup and a control (water)] were used with different solution percentage (25, 50, 75 and 90%). It was observed that honey fed wasp pair produces significantly more number of eggs, adults and also lived significantly longer than other treatments. Highest fecundity and longevity of parasitoid was recorded in case of parasitoids feeding on 50% honey solution followed by 25% sugar and date syrups solutions respectively. It has provided useful information for the mass rearing of this potential biocontrol agent which may be used in the successful implementation of biocontrol program for the suppression of pyralid pest insects damaging different agricultural crops of economic importance.
Article Information
Received 18 September 2015
Revised 23 August 2016
Accepted 24 August 2016
Available online 21 April 2017
Authors’ Contribution
ZA and RSAK designed the project and supervised the work. SA performed the experimental work. FH helped in execution of experimental work. SA and SKA wrote the article. MT helped in rearing of insects. SR helped in collection and management of data. MA helped in handling of experimental insects.
Key words
Bracon hebetor, Fecundity, Longevity, Honey syrup, Date syrup, Sugar syrup, Parasitic wasp, Parasitoid, Galleria mellonella.
DOI: http://dx.doi.org/10.17582/journal.pjz/2017.49.3.761.767
* Corresponding author: zainunibas@gmail.com
0030-9923/2017/0003-0761 $ 9.00/0
Copyright 2017 Zoological Society of Pakistan
INTRODUCTION
Like other insects, parasitoids also require continuous supply of balanced food to perform their activities. Food consumption, especially from sugar rich sources, enhance the reproductive potential and insect parasitoid longevity (Lee and Heimpel, 2005, 2008; Lavandero et al., 2006; Winkler et al., 2009). In the context of biological control program, sugar feeding plays a vital role in the performance of parasitoids (Gurr et al., 2004). For parasitoids various sugars have different nutritional qualities and strongly increased the longevity while the others have small effect or even may be toxic to parasitoids (Wäckers, 2001; Wäckers et al., 2006; Kehrli and Bacher, 2008). Along with enhancing the longevity and fecundity (Heimpel et al., 1997a, b; Wäckers, 2001; Costamagna and Landis, 2004; Winkler et al., 2006), carbohydrate consumption also affects other aspects of reproductive success (Azzouz et al., 2004) in insect parasitoids.
Honey is used as best food source and standard for comparison with other food sources. Malati and Hatami (2005) investigated that feeding on honey significantly enhanced the life span of adults of Trichogramma brassicae Bezdenko. Eliopoulos (2007) determined that adult life span and number of adult emergence in Venturia canescens Gravenhorst (Hymenoptera: Ichneumonidae) was strongly dependent on honey feeding.
Onagbola et al. (2007) studied the life span, reproductive potential and sex ratio of progeny of an ectoparasitoid Pteromalus cerealellae (Ashmead) (Hymenoptera: Pteromalidae) which were affected by feeding on different diets. Wu et al. (2008) showed that Meteorus pulchricornis parasitoids when provided with 30% sugar solution lived significantly longer and produced more progeny compared to those fed on other concentration of sugars in the absence of host. Wyckhuys et al. (2008) checked that Binodoxys communis had more longevity when provided with honey and sucrose as compared to those provided with honeydew of Aphis glycines. Harvey et al. (2012) reported that reproductive potential and longevity in two hyperparasitoids, Lysibia nana and Gelis agilis, showed significant variations when they were provided with different dietary sugars and honey.
Bracon hebetor (Say.) (Hymenoptera: Braconidae), is a cosmopolitan larval ectoparasitoid that utilizes several pests belonging to the family Pyralidae (Lepidoptera) as host (Fagundes et al., 2005; Shojaei et al., 2006; Yasodha and Natarajan, 2006; Desai et al., 2007; Kyoung et al., 2008; Mohapatra et al., 2008; Dweck et al., 2010). The B. hebetor females prefer to attack and oviposit on last instar (fifth) larvae, although younger instars are also parasitized. The broad host range, high reproductive rate and short generation time makes B. hebetor is an excellent natural biological control agent of many pyralid pests, including Galleria mellonella (Lepidoptera: Pyralidae) while no work has been done on the diet preference effects on the reproductive potential and fecundity of this biocontrol agent (Gunduz and Gulel, 2005).
The project was planned to check that if fecundity and longevity of B. hebetor is affected when fed on honey and different sugars and to determine at which diet maximum female emergence is obtained. It is hypothesized that fecundity and longevity of B. hebetor will be affected due to dietary sugars and honey which provide maximum longevity and fecundity compared to other sugars.
MATERIALS AND METHODS
Rearing of host Galleria mellonella
The larvae, pupae and adults of the host, greater wax moth, G. mellonella were collected from the infested bee hives located at the campus of University of Agriculture, Faisalabad, Pakistan. Pairs of adult male and female of G. mellonella were placed in the jars (having fresh wax) for mating. After 24 h mating period, female starts egg laying as egg firing by extruding its ovipositor and flattering wings. Eggs were placed in incubator for hatching under controlled conditions. The insect culture containing eggs, larvae, pupae and adults was maintained at 27ºC±1 temperatures and 65±5% relative humidity (RH) by following a slightly modified approach as described by Anam et al. (2015).
Rearing of Bracon hebetor
The ectophagous larval parasitic wasp B. hebetor was reared in the laboratory on the late stage (5th instar) larvae of greater wax moth G. mellonella by following a slightly modified approach as described by Manzoor et al. (2011) and Anam et al. (2015). The adults of the parasitoid, B. hebetor were collected directly from the berseem crop, Trifolim alexandrium L., located at the campus of the University of Agriculture, Faisalabad, Pakistan. The collected parasitoids were identified on the basis of morphological characters by making comparison with the available literature. The parasitoid cultures was also maintained in glass jars, placed at 27ºC ±1, 65±5% relative humidity (RH) and 18 h light /6 h dark photoperiod.
Experimental procedure
Different concentrations (25%, 50%, 75% and 90%) of pure honey and date syrup were prepared with distilled water in 25ml glass vial. Similarly fine powder of sugar was dissolved in water to make its 25%, 50%, 75% and 90% concentrations in different pre-labeled glass vials of 25 ml. Honey syrup, date syrup and sugar syrup were used as treatments and different concentrations (25%, 50%, 75% and 90%) were used as different levels of these treatments.
Three groups of vials were prepared G1, G2, and G3 for three different diets like honey, sugar and date syrup, respectively. Further, five vials for each group were also prepared and vials for each group were provided with one larva of G. mellonella, a couple of freshly emerged parasitoid males and females. The cotton plugs soaked in four different concentrations (25%, 50%, 75% and 90%) of all the three diets (honey syrup, date syrup and table sugar syrup) were wrapped on the mouth of each vial of each group. The 5th vial was provided with water soaked cotton plug which acted as control. After egg laying, parasitoids were removed from the vials and shifted in other three groups. This practice was repeated again and again till the death of female to check its longevity. The number of eggs laid on each host were recorded after 24 h intervals and then parasitized hosts were incubated individually till adult emergence. The number of eggs hatched, emerged larvae feed on parasitized host larvae and pupate outside the body. The number of adults emerged and their sex ratio were also recorded on daily basis from each vial.
Determination of sex ratio
Sex ratio of B. hebetor and the percentage of female emergence in response to different diets was calculated by using the following formula:
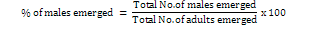
Data analysis
The data was statistically analyzed by Analytical software (2003), Statistix 8.1 by using the factorial design and the means were separated by Tukey HSD test at P<0.05.
RESULTS
Number of egg laying and adult emergence of Bracon hebetor due to different diets
Data given in the Table I show the mean values of fecundity and adult emergence of B. hebetor in response to different diets. Results show that there is a statistically significant difference in fecundity and adult emergence of B. hebetor fed on different concentrations of honey, sugar and date syrup (Tukey’s HSD test, P< 0.05). Parasitoid female fed on 50% honey solution laid significantly more number of eggs (ANOVA, df=3, F=128.22, 129±4, P<0.05) and produced more progeny (107.4±4.91, P<0.05) than those fed on other concentrations of different diets. When sugar and date syrups were provided to parasitoids as food, maximum fecundity (96.4±2.50 and 77±1.94, respectively) and maximum adult emergence (ANOVA, df=3, F=104.09, P<0.05, 85.5±2.85 and 66.4±2.204, respectively) were recorded at 25% concentration but it was still smaller than those which fed on 50% honey solution. Whereas in control treatment average fecundity of 27.1±0.70 (P<0.05) and adult emergence 15.5±0.70 (P<0.05) was recorded.
Sex ratio on different diets
Figure 1 shows the mean percentage of male and female sex ratio emerged by feeding on different diets. The results showed that maximum female emergence (48.4±3.36%, p<0.05) was observed by feeding on 50% honey solution followed by 43±1.74, 43.5±1.51 and 37.5±0.71% after sugar, date syrup and control treatment, respectively. Minimum female percentage (41.1±0.51%, p<0.05) was recorded in date syrup on 90% solution. Male mean percentage was recorded maximum (59.7±1.83%) on 90% honey solution while minimum (51.5±6.02%, p<0.05) was observed on 50% honey solution.
Table I.- Mean effect of different diets on fecundity and adult emergence of Bracon hebetor.
| Treat- ments |
Concentrations |
|||||||
|
25% |
50% |
75% |
90% |
|||||
|
Fecundity |
Adults emerged |
Fecundity |
Adult |
Fecundity |
adult |
Fecundity |
Adult |
|
| Honey |
63.4 ±1.805d |
50.4 ±2.94de |
129 ±4.00 a |
107.4 ±4.91a |
98.4± 2.42 b |
81.6± 4.06 b |
47.8± 1.93ef |
35.8± 2.009f |
| Sugar |
96.4 ±2.50 b |
85.5 ±2.85 b |
65.6 ±3.18cd |
55.4± 2.56cd |
48.4± 3.23ef |
39.6± 2.85ef |
45.0± 2.00 f |
35± 1.41 f |
| Date syrup |
77.0 ±1.94 c |
66.4 ±2.20 c |
58± 3.20 de |
50.6± 2.73de |
44.0± 1.87 f |
34.4± 2.85 f |
42.4± 1.74 f |
32.6± 1.63 f |
| Control |
27.0 ±0.70 g |
15 ±0.70 g |
26.4± 1.69 g |
16± 0.70 g |
27.0± 0.70 g |
15± 0.70 g |
28.0± 0.70 g |
16± 0.70 g |
Means followed by different letter(s) within each column (denoted by lower-case letters) are significantly significant by Tukey’s HSD test at P <0.05.
Longevity of Bracon hebetor on different diets
Figure 2 indicates the mean value of longevity of male and female of B. hebetor in response to different diets. Maximum female and male life span (21.8±0.66 and 13.6±0.24 days, respectively) was recorded when parasitoids fed on 50% honey solution than those fed on other concentrations of honey and of other diets. In case of sugar feeding parasitoids, female showed longer life span of 19.2±0.37 days and male had longer life span of 12.6±0.50 days at 25% concentration. Date syrup feeding female and male parasitoids showed maximum of 15.8±0.37 and 10.6±0.4 days longevity respectively at 25% concentration. More dilutions in the case of all treatments caused decline in the longevity of parasitoids.
DISCUSSION
Several studies have shown that the survival, longevity, sex ratio and fecundity of parasitoids largely depend upon diet. Many authors have reported that supply of an artificial source of food is very important and beneficial for the use of parasitic wasps in agricultural systems (Heimpel et al., 1997a).
Our results show that different solutions of honey, sugar and date syrup have significant effect on longevity, fecundity and female emergence of B. hebetor. Parasitoids fed on 50% honey solution lived relatively longer and deposited more number of eggs than those fed on all other solutions. This indicated that 50% honey was a better diet for B. hebetor, its longevity and fecundity decreased with increase or decrease in honey concentration. This was probably due to the sugar and water content of the honey (Barbehenn et al., 1999). Lower concentration of honey contains more water than carbohydrates while higher concentration of honey contains more energy than water, i.e. too much energy or lack of water which will shorten the parasitoid longevity. Similar results were reported by Leatemia et al. (1995) in which the longevity of females Trichogramma minutum Riley (Hymenoptera: Trichogrammatidae) fed on 20% honey was 26.5±5.4 days, on 40% honey it was 28.5±5.4 days, on 60% honey it was 25.4±6.1 days and on 80% honey it was at 20.7±6.1 days. Temerak (1983) showed that best diet provided to parasitoids to elongate their life span was honey. After honey, maximum egg laying, female emergence and longevity of B. hebetor was recorded after feeding on sugar followed by date syrup.
It was interesting to note that honey, sugar and date syrup solution caused the parasitoids to produce more eggs and also allowed them to live longer. Similar results were reported for Meteorus pulchricornis (Wesmael) in which the sugar concentration maximized lifespan as well as lifetime progeny production (Wu et al., 2008). In addition, Karimi and Hatami (2010) reported that fecundity of honey fed adults increased approximately four folds compared to unfed females. Adults fed on honey showed highest percent emergence over unfed females. Paraiso et al. (2011) recommended provision of honey to Trichogramma females in mass rearing programs to establish a sustainable population of T. fuentesi in the laboratory. It was also reported that mean time to 50% survival of unfed parasitoids T. platneri, was 2.0±0.1 days, whereas honey-fed parasitoids lived significantly longer (2.6±0.1days) (Mansfield and Mills, 2002). Our findings also showed that honey fed parasitoids lived longer than those provided with water only. Figure 1 showed that male emergence in all the treatments were more as compared to females. Kuhlmann and Mills (1999) also reported that females of Trichogramma species with longer longevity are known to produce more males, probably, due to sperm depletion. But sex ratio remained the same (more number of males than females) in all treatments which are in contrast to Onagbola et al. (2007) who reported that feeding on sugar-rich foods alter the sex ratio of progeny.
The composition and concentration of sugars may affect the longevity and number of offspring of parasitoids (Fuchsberg et al., 2007; Wu et al., 2008), but the effects may differ among parasitoid species (Lee et al., 2004; Chen and Fadamiro, 2006; Wyckhuys et al., 2008). Our findings also showed that as the concentrations of honey, sugar and date syrup change, longevity and number of adults emerged also change (Table I).
The present results are mostly in accordance with the above reported findings as mentioned by Harvey et al. (2012) that wasps fed on glucose, as well as on a honey imitating solution that contained approximately the same sugars in similar relative concentrations found in honey, produced only half of the progeny compared to wasps fed on honey. Shaw (1997) stated that honey is an efficient diet for parasitic wasps as it is for bees. It contains proteins and vitamins as well as sugar and seems to be adequate for egg maturation for most, possibly all species that nourish and mature their eggs successively through their adult life.
CONCLUSION
It may be concluded that 50% honey solution was found to be a better food than other concentrations of honey or other diets (sugar, date syrup and control) evidenced that it relates to parasitoid longevity and fecundity increment. A useful information has been generated which may be exploited in mass rearing programmes of the wasp B. hebetor for successful implementation of biocontrol strategies for the suppression of pest insects of Pyralidae
ACKNOWLEDGEMENT
We are grateful to HEC (Higher Education Commission) Government of Pakistan for providing financial support (No. 20-3247/NRPU/R&D/HEC/ 14/408, 25.11.2015) and IFS grant (F/5089-1)
Statement of conflict of interest
Authors have declared no conflict of interest.
REFERENCES
Anam, J., Zain ul Abdin, Arshad, M., Falabella, P., Abbas, S.K., Tahir, M., Jamil, A., Manzoor, A. and Shaina, H., 2015. Multiple effects of host density on egg density and the sex ratio of progeny of Bracon hebetor (Say.) (Hymenoptera: Braconidae). Pakistan J. Zool., 47: 455-460.
Azzouz, H., Giordanengo, P., Wackers, F.L. and Kaiser, L., 2004. Effects of feeding frequency and sugar concentration on behavior and longevity of the adult aphid parasitoid: Aphidius ervi (Haliday) (Hymenoptera: Braconidae). Biol. Contr., 31: 445–452. https://doi.org/10.1016/j.biocontrol.2004.07.013
Barbehenn, R.V., REESE, J.C. and Hagen, K.S., 1999. The food of insects. In: Ecological entomology (eds C.B. Huffaker and A.P. Gutierrez). John Wiley and Sons, New York.
Benson, J.F., 1973. The biology of Lepidoptera infesting stored products, with special reference to population dynamics. Biol. Rev., 48: 1-26. https://doi.org/10.1111/j.1469-185X.1973.tb01113.x
Chen, L. and Fadamiro, H.Y., 2006. Comparing the effects of five naturally occurring monosaccharide and oligosaccharide sugars on longevity and carbohydrate nutrient levels of a parasitic phorid fly, Pseudacteon tricuspis. Physiol. Ent., 31: 46–56. https://doi.org/10.1111/j.1365-3032.2005.00484.x
Costamagna, A.C. and Landis, D.A., 2004. Effect of food resources on adult Glyptapanteles militaris and Meteorus communis (Hymenoptera: Braconidae), parasitoids of Pseudaletia unipuncta (Lepidoptera: Noctuidae). Environ. Ent., 33: 128-137. https://doi.org/10.1603/0046-225X-33.2.128
Desai, V.S., Nagwekar, D.D., Patil, P.D. and Narangalkar, A.L., 2007. Field evaluation of a larval parasite Bracon hebetor Say against coconut black headed caterpillar. J. Pl. Crops, 35: 188-189.
Dweck, H.K.M., Glenn, P.S., Gunduz, E.A. and Anderbrant, O., 2010. Kairomonal response of the parasitoid, Bracon hebetor (Say.) to the male-produced sex pheromone of its host, the greater wax moth, Galleria mellonella (L.). J. chem. Ecol., 36: 171–178. https://doi.org/10.1007/s10886-010-9746-x
Eliopoulos, P.A., 2007. The importance of food supplements for parasitoids of stored product pests: the case of Venturia canescens (Hymenoptera: Ichneumonidae). Integ. Protec. Stored Prod. IOBC/wprs Bull., 30: 37-41.
Fagundes, G.G., Mohamed, H.H. and Solis, D.R., 2005. Biological responses of Anagasta kuehniella and its parasitoid, Bracon hebetor, to microwaves radiation (2450 MHz). Rev. Agric. Piracicaba, 80: 12-34.
Fuchsberg, J.R., Yong, T.H., Losey, J.E., Carter, M.E. and Hoffmann, M.P., 2007. Evaluation of corn leaf aphid (Rhopalosiphum maidis; Homoptera: Aphididae) honeydew as a food source for the egg parasitoid Trichogramma ostriniae (Hymenoptera: Trichogrammatidae). Biol. Control, 40: 230-236. https://doi.org/10.1016/j.biocontrol.2006.10.009
Gurr, G. M., Wratten, S.D. and Altieri, M. A., 2004. Ecological engineering for pest management: Advances in habitats manipulation for arthropods. CABI Publishing, Wallingford, pp. 232.
Gunduz, E.A. and Gulel, A., 2005. Investigation of fecundity and sex ratio in the parasitoid Bracon hebetor Say (Hymenoptera: Braconidae) in relation to parasitoid age. Turk. J. Zool., 29: 291-294.
Harvey, J.A., Cloutier, J., Visser, B., Ellers, J., Wäckers, F.L. and Gols, R., 2012. The effect of different dietary sugars and honey on longevity and fecundity in two hyperparasitoid wasps. J. Insect Physiol., 58: 816–823. https://doi.org/10.1016/j.jinsphys.2012.03.002
Heimpel, G.E., Rosenheim, J.A. and Kattari, D., 1997a. Adult feeding and lifetime reproductive success in the parasitoid Aphytis melinus. Ent. Exp. Appl., 83: 305– 315. https://doi.org/10.1046/j.1570-7458.1997.00185.x
Heimpel, G.E., Antolin, M.F., Franqui, R. and Strand, M.R., 1997b. Reproductive isolation and genetic variation between two ‘‘strains’’ of Bracon hebetor (Hymenoptera: Braconidae). Biol. Contr., 9: 149–156. https://doi.org/10.1006/bcon.1997.0529
Karimi, A.M. and Hatami, B., 2010. Effect of feeding and male presence on some biological characteristics of female Trichogramma brassicae (Hymenoptera: Trichogrammatidae). J. Ent. Soc. Iran, 29: 1-11.
Kehrli, P. and Bacher, S., 2008. Differential effects of flower-feeding in an insect host- parasitoid system. Basic appl. Ecol., 9: 709–717. https://doi.org/10.1016/j.baae.2007.07.001
Kuhlmann, U. and Mills, N.J., 1999. Comparative analysis of the reproductive attributes of three commercially produced Trichogramma species and the influence of parasitoid size. Biocontr. Sci. Technol., 9: 335–346. https://doi.org/10.1080/09583159929596
Kyoung, D.J., Ha, D.H., Nho, S.K., Song, K.S. and Lee, K.Y., 2008. Up regulation of heat stock protein genes by envenomation of ectoparasitoid Bracon hebetor in larval host of Indian meal moth, Plodia interpunctella. J. Inverteb. Pathol., 97: 306-309. https://doi.org/10.1016/j.jip.2007.10.001
Lavandero, B.I., Wratten, S.D., Didham, R.K. and Gurr, G.M., 2006. Increasing floral diversity for selective enhancement of biological control agents: a double-edged sward? Basic appl. Ecol., 7: 236–243. https://doi.org/10.1016/j.baae.2005.09.004
Leatemia, J.A., Laing, J.E. and Corrigan, J.E., 1995. Effects of adult nutrition on longevity, fecundity and offspring sex ratio of Trichogramma minutum Riley (Hymenoptera: Trichogrammatidae). Can. Entomol., 127: 245-254. https://doi.org/10.4039/Ent127245-2
Lee, J.C. and Heimpel, G.E., 2005. Impact of flowering buckwheat on Lepidopteran cabbage pests and their parasitoids at two spatial scales. Biol. Control, 34: 290–301. https://doi.org/10.1016/j.biocontrol.2005.06.002
Lee, J.C. and Heimpel, G.E., 2008. Effect of floral nectar, water, and feeding frequency on Cotesia glomerata longevity. Biol. Control, 53: 289–294. https://doi.org/10.1007/s10526-007-9070-8
Lee, J.C., Heimpel, G.E. and Leibee, G.L., 2004. Comparing floral nectar and aphid honeydew diets on the longevity and nutrient levels of a parasitoid wasp. Ent. exp. Appl., 111: 189–199.
Malati, A.K. and Hatami, B., 2005. Effect of honey, sugar and protein on longevity of Trichogramma brassicae (Hymenoptera: Trichogrammatidae) with and without host eggs. J. Ent. Soc. Iran, 25: 1-12.
Mansfield, S. and Mills, N.J., 2002. Direct estimation of the survival time of commercially produced adult Trichogramma platneri Nagarkatti (Hymenoptera: Trichogrammatidae) under field conditions. Biol. Control, 25: 41-48. https://doi.org/10.1016/S1049-9644(02)00027-0
Manzoor, A., Zain-ul-Abdin, Arshad, M., Gogi, M.D., Shaina, H., Mubarik, E., Abbas, E.K. and Khan, M.A., 2011. Biological activity of the toxic peptides from venom of Bracon hebetor (Say) (Hymenoptera: Braconidae). Pak. Entomol., 33: 125-130.
Mohapatra, S.D., Duraimurugan, P. and Saxena, H., 2008. Natural parasitization of Maruca vitrata (Geyer) by Bracon hebetor Say. Pulses Newsl., 19: 11.
Onagbola, E.O., Fadamiro, H.Y. and Mbata, G.N., 2007. Longevity, fecundity, and progeny sex ratio of Pteromalus cerealellae in relation to diet, host provision, and mating. Biol. Contr., 40: 222-229. https://doi.org/10.1016/j.biocontrol.2006.10.010
Paraiso, O., Hight, S.D., Karimo, M.T.K. and Bloem, S., 2011. Egg parasitoids attacking Cactoblastis cactorum (Lepidoptera: Pyralidae) in North Florida. Florida Entomol., 94: 81-90. https://doi.org/10.1653/024.094.0111
Shaw, M.R., 1997. Rearing parasitic hymenoptera. Amat. Entomol., 25: 1-46.
Shojaei, S., Safaralizadeh, M. and Shayesteh, N., 2006. Effect of temperature on the functional response of Habrobracon hebetor Say to various densities of the host, Plodia interpunctella (Hubner). Pak. Entomol., 28: 51-55.
Statistix, 8.1. 2003. User’s manual. Analytical software. Tallahasee, Florida.
Temerak, S.A., 1983. Longevity of Bracon brevicornis (Hym: Braconidae) adults as influenced by nourishment on artificial and natural foods. Entomophaga, 28: 145-150. https://doi.org/10.1007/BF02372138
Wäckers, F.L., 2001. A comparison of nectar- and honeydew sugars with respect to their utilization by the hymenopteran parasitoid Cotesia glomerata. J. Insect Physiol., 47: 1077-1084. https://doi.org/10.1016/S0022-1910(01)00088-9
Wäckers, F.L., Lee, J.C., Heimpet, G.E., Winkler, K. and Wagenaar, R., 2006. Hymenopteran parasitoids synthesize honeydew specific oligosaccharides. Functionl. Ecol., 20: 790-798. https://doi.org/10.1111/j.1365-2435.2006.01158.x
Winkler, K., Wäckers, F.L., Bukovinzski, K.G. and Lenteren, J.C.V., 2006. Sugar resources are vital for Diadegma semiclausum fecundity under field conditions. Basic appl. Ecol., 7: 133-140. https://doi.org/10.1016/j.baae.2005.06.001
Winkler, K., Wäckers, F.L., Kaufman, L.V., Larraz, V. and Lenteren, J.C.V., 2009. Nectar exploitation by herbivores and their parasitoids is a function of flower species and relative humidity. Biol. Control, 50: 299–306. https://doi.org/10.1016/j.biocontrol.2009.04.009
Wu, H., Meng, L. and Li, B., 2008. Effects of feeding frequencies and sugar concentrations on lifetime reproductive success of Meteorus pulchricornis (Hymenoptera: Braconidae). Biol. Control, 45: 353-359. https://doi.org/10.1016/j.biocontrol.2008.01.017
Wyckhuys, K.A.G., Strange-George, J.E., Kulhanek, C.A., Wäckers, F.L. and Heimpel, G.E., 2008. Sugar feeding by the aphid parasitoid Binodoxys communis: How dose honeydew compare with other sugar sources. J. Insect Physiol., 54: 481-491. https://doi.org/10.1016/j.jinsphys.2007.11.007
Yasodha, P. and Natarajan, N., 2006. Diversity of natural enemies of Leucinodes orbonalis Guenee. Entomon, 31: 323-326.
To share on other social networks, click on any share button. What are these?










