Volumetric and Volume Fractional Comparison of the Nasal Structures of the Stork (Ciconia ciconia) and Seagull (Larus fuscus) Using Computed Tomography Images
Volumetric and Volume Fractional Comparison of the Nasal Structures of the Stork (Ciconia ciconia) and Seagull (Larus fuscus) Using Computed Tomography Images
Burcu Onuk1,*, Murat Kabak1, Bünyamin Sahin2 and Nazan Gezer İnce3
1Department of Anatomy, Faculty of Veterinary Medicine, Ondokuz Mayıs University, Samsun, Turkey
2Department of Anatomy, Medical Faculty, Ondokuz Mayıs University, Samsun, Turkey
3Department of Anatomy, Faculty of Veterinary Medicine, Istanbul University, İstanbul, Turkey
ABSTRACT
The rostral part of the nasal cavity is cooler to prevent the loss of water that is crucial for the fluid balance of birds. This function may be related to the structure of the nose of flying birds. For this reason, we decided to analyze the nasal structure of two different birds using volume and volume fraction approach of the stereology. In this study, 14 seagulls and 7 storks were examined and volume fractions of the nasal cavity and nasal structures were estimated. Heads of animals were scanned in computed tomography. The images were sampled randomly at a 1/5 sampling fraction. 11-16 sections for the stork and 16-20 sections for the seagull were examined. The sectional surface areas of the total nasal cavity, nasal septum, nasal space and conchae were estimated using the Cavalieri principle of the stereological methods. The volume and volume fractions of structures were assessed. Results obtained from the CT images were compared between stork and seagull. The volume fractions of the conchae, nasal cavity and nasal septum were 30.24%, 55.22%, 14.54%, and 33.03%, 53.23%, 13.73%, in stork and seagulls, respectively. The volume fractions of structures did not show statistical difference between two species. Our findings suggest that the nasal structures of the two-different species have similar architecture, which may be linked to the same function in flying birds.
Article Information
Received 13 June 2015
Revised 23 May 2017
Accepted 30 August 2017
Available online 18 April 2018
Authors’ Contribution
BO and MK designed the study. BO and BS performed sterological analysis. BO and NGI collected samples. BO, MK, BS and NGI wrote the article.
Key words
Cavalieri principle, Computed tomography, Nasal cavity, Seagull, Stork.
DOI: http://dx.doi.org/10.17582/journal.pjz/2018.50.3.963.967
* Corresponding author: [email protected]
0030-9923/2018/0003-0963 $ 9.00/0
Copyright 2018 Zoological Society of Pakistan
INTRODUCTION
The anterior part of the nasal cavity, which is important in preserving water and energy, is colder than the posterior part in avian species. Due to this temperature difference, loss of water is prevented by regaining water from the humid air during exhalation. Saving water this way is particularly important for migratory birds (Getty, 1975; Ocal and Erden, 2002). There are large venous plexuses in the lamina propria of the conchae located in the nasal cavity. These plexuses are filled with blood in every 20-30 min and swell the nasal mucosa so that the airflow is reduced. These structures increase in surface area and warm the inhaled air by changing the direction of airflow (Ahishali et al., 2006).
Cavalieri principle is one of the methods used in stereology for calculation of volume and volume fraction (Diab et al., 1998). Reliable and objective numerical results can be obtained by using this method (Glaser and Glaser, 2000) that was developed by Italian mathematician Bonaventura Cavalieri for estimating the volume of 3-dimentional subjects (Roberts et al., 1997; Odaci et al., 2003). Effectiveness of the method has been proven in a number of studies (Calmon and Roberts, 2000; Roberts et al., 2000; Onuk et al., 2013).
Physiological and anatomical structures of the nose have been studied by Grutzenmacher et al. (2011). Geometrical measurements of the nasal cavity were made by using acoustic rhinometry and computed tomography in human (Gilain et al., 1997), in guinea pig, and rat (Straszerk and Pedersen, 2004). Despite the extensive literature survey, no data regarding this region has been found in any species except for domestic goose (Onuk et al., 2013). For this reason, we decided to analyze the nasal structure of two different birds (migratory and non- migratory birds) using volume fraction approach of stereology that gives quantitative data independent of the body sizes of the examined subjects.
Materials and methods
In the present study, 1040-1490 g live weight adult 14 seagulls (Larus fuscus), a non- migratory bird from Schizognath group, that were used in different studies in the Department of Anatomy, Faculty of Veterinary Medicine, Istanbul University and 3480-3920g live weight seven adult storks (Ciconia ciconia), migratory birds from Desmognath group, when they were about to die and could not be saved were used for the study. In this study, materials that were free of respiratory diseases and pathological symptoms were observed. The study was carried out with the permission of the General Directorate of Nature Conservation and National Parks of the Ministry of Forestry and Water Affairs. Nasal cavities of animals were scanned using computerized tomography (CT) with transverse orientation (Fig. 1).
The 512x512 sized images with 0.5 mm thickness were obtained using Toshiba Aquilion 16 computed tomography. The images were stored as DICOM files and transferred to a CD. Using the ImageJ software, the images were sampled randomly with 1/5 fraction (a program distributed freely by the National Institute of Health of the USA). Depending on the length of nose, 11-16 sections for the stork and 16-20 sections for the seagull were examined (Fig. 1A, B in both top and bottom panels). Surface areas of the conchae, nasal septum and nasal cavity were estimated using the grid plugin of the ImageJ software. For this purpose, grids with representing areas of 0.3 and 0.4 mm2 were randomly thrown onto sections and the number of plusses hitting the interested structure were counted (Fig. 1C in both top and bottom panels).
The volumes were estimated by multiplication of representing areas of grids with the total number of points hitting the structure as described before (Onuk et al., 2013). The total volume of nasal cavity is assessed using the polygon selection toll of the ImageJ. The borders of the nasal cavity were drawn in all sections (Fig. 1B, C in both top and bottom panels) and total volume of the nasal cavity was calculated by multiplying total surface area with the section thickness. The volume fractions of the conchae and nasal cavity within the total nasal cavity were calculated using the equation:
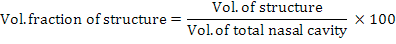
To assess whether point intervals on the grid and the number of sections were adequate, the coefficient of error (CE) were calculated (Gundersen and Jensen, 1987). The volumes, volume fractions and CE values were calculated automatically by entering the number of points, representing areas per point and sectional surface areas in an excel worksheet of Microsoft.
Data obtained from CT section images were evaluated for the data structure and GENMOD (Generalized Linear Modal) procedure was used to compare the methods using SAS (2006).
Results
In this study, volume (cm3) and volume fractions (%) of structures located in the nasal cavity of seagull and of stork were calculated by applying the Cavalieri Principle to the images obtained from CT images. The mean volumes of the total nasal cavity, conchae, nasal space and nasal septum were recorded as 3.89 cm3, 1.27 cm3, 2.09 cm3, 0.53 cm3 for the seagull and 4.36 cm3, 1.32 cm3, 2.41 cm3, 0.63 cm3 for the stork, respectively (Table I). In addition, values of volume fractions for conchae, nasal space and nasal septum were determined as 33.03%, 53.23%, 13.73% for the seagull and 30.24%, 55.22%, 14.54% for the stork, respectively (Table I). Finally, all the values of volume and volume fraction were compared. The mean volumes of the total nasal cavity, conchae, nasal space and nasal septum were slightly larger in the stork than that of the seagull. However, the differences did not rise to a statistically significant level (p≥ 0.05).
Discussion
Information about the data of the normal anatomical structures of living creatures is of importance in accurate evaluation of radiographic examinations, in understanding of the surgical procedure results (Weiglein et al., 1992), and in the diagnosis of diseases widespread among avian species such as avian influenza and laryngotracheitis (Calnek et al., 1991).
Anatomical structures are used in taxonomical differentiation of avian species. Based on the skull, birds are divided into four (4) groups as palaeognath, schizognath, desmognath and aegithognath (Demirsoy, 1992).
The first study on nasal cavity and size of conchae of avian species was carried out on domestic goose by Onuk et al. (2013). Standard values were obtained by comparing the physical sections and CT images. In addition, it has been reported that compared to the physical sections, the sections obtained from CT images better represent the real values with very minor deviation because of not being affected by the partial voluming effect depending on the section thickness (Sahin et al., 2003; Onuk et al., 2013). Therefore, CT was preferred to investigate the nasal cavity and conchae of the seagull and stork.
Table I.- Descriptive data for the volume (cm3) of structures and volume fractions (%) of structures located in the nasal cavity of seagull and stork.
| Measurement |
Seagull |
Stork |
||
|
Volume±SD (cm3) |
Min-Max |
Volume±SD (cm3) |
Min-Max |
|
|
Structures in the nasal cavities |
||||
| Total volume of nasal cavity |
3.89±0.13 |
3.12-4.53 |
4.36±0.31 |
3.56-5.46 |
| Conchae volume |
1.27 ±0.04 |
0.95-1.60 |
1.32±0.15 |
0.83-2.15 |
| Space volume |
2.09±0.10 |
1.41-2.63 |
2.41±0.20 |
1.84-3.20 |
| Nasal septum volume |
0.53±0.03 |
0.35-0.75 |
0.63±0.06 |
0.48-0.88 |
| Volume fractions of structures (%) | ||||
| Conchae fraction |
33.03±1.42 |
22.33-40.54 |
30.24±2.48 |
22.33-5.46 |
| Space fraction |
53.23± 1.28 |
45.03-62.20 |
55.22±2.44 |
44.53-64.83 |
| Nasal septum fraction |
13.73± 0.54 |
10.99-17.57 |
14.54±1.03 |
11.62-19.49 |
The volume and volume fraction of nasal cavity structures of the two different bird species that are taxonomically different has been determined for the first time in the current study. The results showed that these two species cannot be differentiated based on the parameters investigated in this study because of high similarity. It has been reported in literature (Getty, 1975; Ocal and Erden, 2002) that rostral part of the nasal cavity of migratory birds was colder compared to the caudal part, and the water was saved by means of this temperature difference. If the conchae volume and the nasal temperature are lower, the water loss is lesser. According to this principle, it was expected that the respective volume of the conchae in the migratory birds was lower. But, it is viewed that the conchae volumes and volume fractions were not important in preserving water, because there was no difference between the two species. Also, Ahishali et al. (2006) reported the presence of large plexus which increase the humidity and warmth in the expiration of air. These plexuses cause water loss by evaporating humidity in the exhaled air. If the number of plexus is less, water loss in the nasal cavity is expected to be at the minimum level. Due to lack of a significant difference between the conchae volumes and volume fractions of stork and seagull, we inferred that there will be less amount of plexus in stork’s conchae than in seagull’s conchae.
For better understanding of water preservation in migratory birds, more detailed studies about the conchae venous plexus density and the morphologic structure of the conchae epithelium are required. The first data on the nasal cavity and the conchae volume/volume fractions in the stork and seagull, contributes to the anatomy literature.
Acknowledgements
This study was supported by Project Management Office of Ondokuz Mayıs University (Project No: PYO.-VET.1901.11.026). The study was presented in the 1st Asian and African Stereology Congress (Samsun, Turkey 2012). The authors are grateful to Dr. Serhat Arslan, from Ondokuz Mayıs Universty, Faculty of Veterinary Medicine, Department of Biometry, for statistical analysis of the data sets.
Statement of conflict of interest
Authors have declared no conflict of interest.
References
Ahishali, B., Arda, O., Aytekin, Y., Dagdeviren, A., Gursoy, E., Sırmalı, S., Solakoglu, S., Tasyurekli, M. and Ugur, Y., 2006. Basic Histology. Nobel Tıp Kitabevleri, Turkey.
Calmon, G. and Roberts, N., 2000. Automatic measurement of changes in brain volume on consecutive 3D MR images by segmentation propagation. Magn. Reson. Imaging, 18: 439-453. https://doi.org/10.1016/S0730-725X(99)00118-6
Calnek, B.W., Barnes, H.J., Beard, C.W., Reid, W.M. and Yoder, H.W., 1991. Diseases of poultry. Iowa State University Press, Iowa.
Demirsoy, A., 1992. Vertebrates (Reptiles, Aves and Mammalians): Basic Rules of Life. Meteksan A.S, Ankara.
Diab, K.M., Ollmar, S., Sevastik, J.A., Willes, U., Svensson, A., 1998. Volumetric determination of normal and scoliotic vertebral bodies. Eur. Spine J., 7: 282-288. https://doi.org/10.1007/s005860050076
Getty, R., 1975. Sison and Grossman’s. The anatomy of the domestic animals. WB Saunders Company, Philadelphia.
Gilain, L., Coste, A., Rieolfi, F., Dahan, E., Marlıae, D., Peynegre, R., Harf, A. and Louis, B., 1997. Nasal cavity geometry measured by acustic rhinometry and computed tomography. Arch. Otolaryngol. Head Neck Surg., 123: 401-405. https://doi.org/10.1001/archotol.1997.01900040037006
Glaser, J.R. and Glaser, E.M., 2000. Stereology, morphometry and mapping: the whole is greater than the sum of its parts. J. Chem. Neuroanat., 20: 115-126. https://doi.org/10.1016/S0891-0618(00)00073-9
Grutzenmacher, S., Robinson, D.M., Sevecke, J., Mlynski, G. and Beule, A.G., 2011. Comparative investigations of anatomy and physiology in mammalian noses (Homo sapiens, Artiodactyla). Rhinology, 49: 18-23.
Gundersen, H.J. and Jensen, E.B., 1987. The efficiency ofsystematic sampling in stereology and its prediction. J. Microscop., 147: 229-263. https://doi.org/10.1111/j.1365-2818.1987.tb02837.x
Odaci, E., Sahin, B., Sonmez, O.F., Kaplan, S., Bas, O., Bilgic, S., Bek, Y. and Ergur, H., 2003. Rapid estimation of the vertebral body volume: A combination of the Cavalieri principle and computed tomography images. Eur. J. Radiol., 48: 316-326. https://doi.org/10.1016/S0720-048X(03)00077-9
Ocal, K. and Erden, H., 2002. The anatomy of the domestic birds. Medisan Press, Ankara..
Onuk, B., Kabak, M., Sahın, B., Ince, N.G. and Selcuk, M.B., 2013. New method for estimating the volume and volume fractions of the nasal structures in the goose (Anser anser domesticus) using computed tomography images. Br. Poult. Sci., 54: 441-446. https://doi.org/10.1080/00071668.2013.806980
Roberts, N., Cruz-Orive, L.M., Bourne, M., Herfkens, R.J., Karwoskis, R.A. and Whitehouse, G.H., 1997. Analysis of cardiac function by MRI and stereology. J. Microscop., 187: 31-42. https://doi.org/10.1046/j.1365-2818.1997.2040764.x
Roberts, N., Puddephat, M.J. and Mcnulty, V., 2000. The benefit of stereology for quantitative radiology. Br. J. Radiol., 73: 679-697. https://doi.org/10.1259/bjr.73.871.11089458
Sahin, B., Emırzeoglu, M., Uzun, A., Incesu, L., Bek, Y., Bilgic, S. and Kaplan, S., 2003. Unbiased estimation of the liver volume by the Cavalieri principle using magnetic resonance images. Eur. J. Radiol., 47: 164-170. https://doi.org/10.1016/S0720-048X(02)00152-3
SAS, 2006. SAS Statistical Software ver. 9.1.3. SAS Inst. Inc., SAS Campus Drive, Cary, North Caroline, 27513, Cary, NC, USA.
Straszek, S.P. and Pedersen, O.F., 2004. Nasal cavity dimensions in guinea pig and rat measured by acoustic rhinometry and fluid-displacement method. J. appl. Physiol., 96: 2109-2114. https://doi.org/10.1152/japplphysiol.00540.2003
Weiglein, A., Anderhuber, W. and Wolf, G., 1992. Radiologic anatomy of the paranasal sinus in the child. Surg. Radiol. Anat., 14: 335-339. https://doi.org/10.1007/BF01794761
To share on other social networks, click on any share button. What are these?










