Purification and Characterization of a Thermostable Pyruvate Ferredoxin Oxidoreductase/Pyruvate Decarboxylase from Thermococcus kodakaraensis
Purification and Characterization of a Thermostable Pyruvate Ferredoxin Oxidoreductase/Pyruvate Decarboxylase from Thermococcus kodakaraensis
Kanwal Nisa1, Sadaf Ashraf1, Masood Ahmed Siddiqui1*,Naila Taj1, Habib-Ur-Rehman1, Arifa Bano1 and Naeem Rashid2
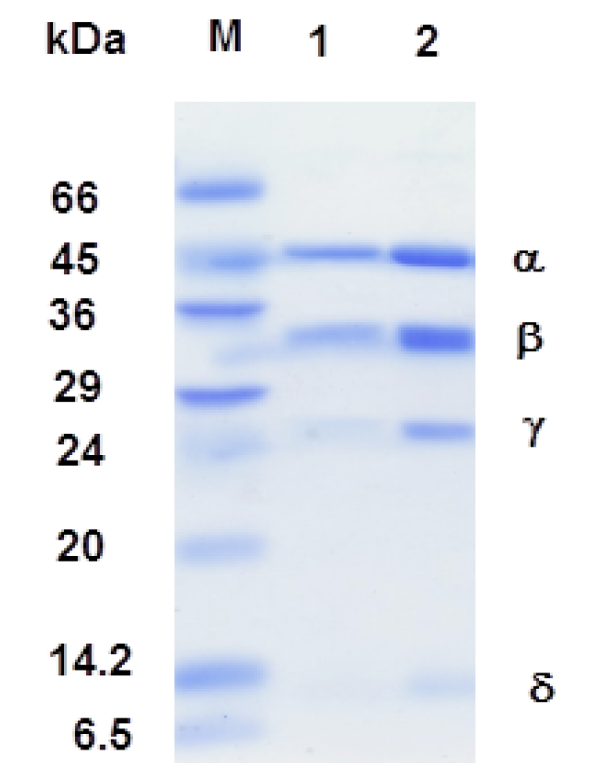
The 0.1% SDS-12.5 polyacrylamide gel electrophoresis. Sample were prepared by boiling for 3 min in a sample buffer (50 mM Tris-HCl pH 6.8, 2% SDS, 0.1% bromophenol blue, 10% glycerol. M: molecular weight markers (Albumin bovine serum, 66,000; ovalbumin, 45,000; glyceraldehydes-3-phosphate dehydrogenase, 36,000; carbonic anhydrase 29,000; trypsinogen 24,000; trypsin inhibitor, 20,000; α-lactalbumin, 14,200 and Aprotenin, 6,500).
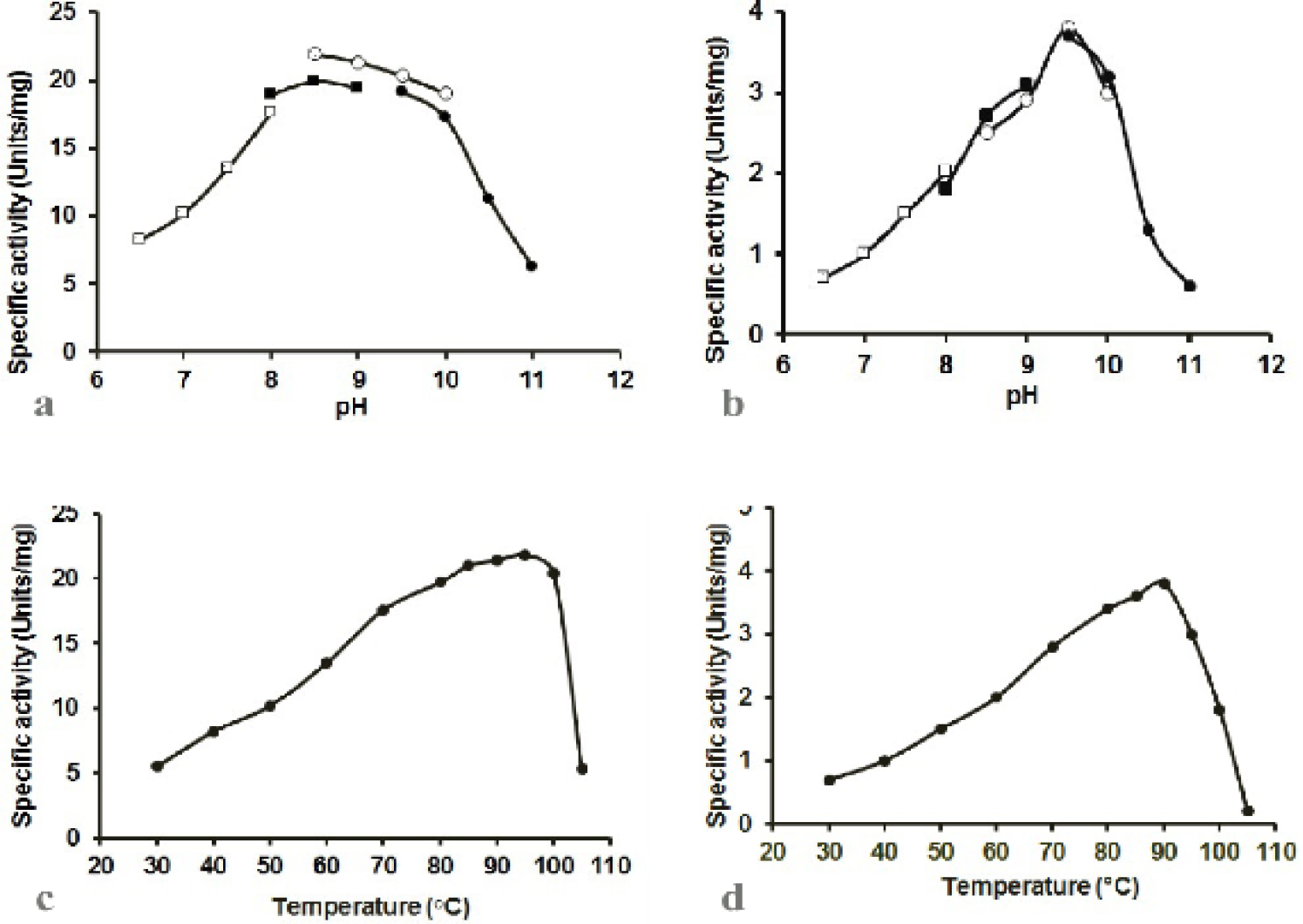
The effect of pH on (a) POR and (b) PDC specific activities of the purified enzyme from T. kodakaraensis. The open boxes indicate the pH dependent experiments with sodium phosphate buffer (pH 6.5, 7.0, 7.5 and 8.0), the closed boxes represent the experiments with Tris HCl buffer (pH 8.0, 8.5 and 9.0), open circle represent the reactions with CHES buffer (pH 8.5, 9.0, 9.5 and 10.0) and closed circles exhibit reactions with CAPS buffer (pH 9.5, 10.0, 10.5 and 11.0). Effect of temperature on (c) POR and (d) PDC specific activities of the purified enzyme from T. kodakaraensis. The assay was performed as described in Materials and method section.
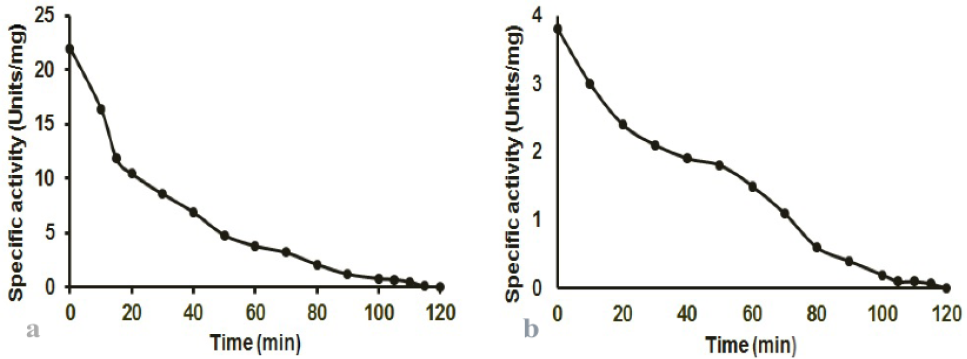
Effect of aerobic conditions on T. kodakaraensis (a) POR and (b) PDC activities. To determine the effect of oxygen on POR and PDC activities, the enzyme was incubated with shaking in the presence of air at 25°C, aliquots were taken at 5-10 min intervals and activity was determined under strict anaerobic conditions.