Effect of NaCl and pH on Bioelectricity Production from Vegetable Waste Extract Supplemented with Cane Molasses in Dual Chamber Microbial Fuel Cell
Effect of NaCl and pH on Bioelectricity Production from Vegetable Waste Extract Supplemented with Cane Molasses in Dual Chamber Microbial Fuel Cell
Muhammad Mohsin Javed1*, Muhammad Azhar Nisar2 and
Muhammad Usman Ahmad2
1Virtual University of Pakistan, M.A Jinnah Campus, Defence Road, Off Raiwind Road, Lahore-Pakistan
2Institute of Industrial Biotechnology, GC University Lahore-54000, Pakistan
ABSTRACT
Vegetable waste has been analyzed for bioelectricity production in locally designed U-shaped microbial fuel cell. These fuel cells were subjected to different concentrations of NaCl and pH for evaluation of power generation. The MFC with 50 mM NaCl concentration showed maximum response and gave 88.9 W/m2 power densities and 569 mV of open circuit voltage. The results showed that NaCl, at its optimum level, plays an important role in increasing the bioelectricity and sustaining the internal resistivity. Six different pH parameters were subjected to check the effectiveness of the treatment on the power production. The maximum power production achieved at pH 4.5 i.e. 70.5 W/m2 of power density at external load of 200 Ω. Which reveals that if the pH difference between the two chambers is increased the power density increases. Bioelectricity production from molasses has not been studied considerably. Present study describes the use of molasses with different organic loads in MFC-1, MFC-2 and MFC-3 with 10, 15 and 20% of sugar contents, respectively, for bioelectricity production. These MFCs were designed locally and maximum power densities of 72820mW/m2, 104400mW/m2and 44340mW/m2for MFC-1, MFC-2 and MFC-3, respectively were obtained at 400Ω external load. The columbic efficiencies were 73.9%, 81.7%and 63.2%, respectively.
Article Information
Received 11 June 2018
Revised 22 July 2019
Accepted 20 November 2020
Available online 12 March 2021
(early access)
Published 03 December 2021
Authors’ Contribution
MMJ supervised the experiemntal work. MAN and MUA performed the experimental work. MMJ and MUA wrote the manuscript.
Key words
Vegetable extract, Microbial fuel cell (MFC), Bioelectricity, Cation exchange membrane, Acidophilic and alkaliphilic
DOI: https://dx.doi.org/10.17582/journal.pjz/20180611050622
* Corresponding author: mmj_bot@yahoo.com, dqe@vu.edu.pk
0030-9923/2022/0001-0247 $ 9.00/0
Copyright 2022 Zoological Society of Pakistan
INTRODUCTION
Microbial fuel cells (MFCs) are the alternative source of energy which is gaining attention day by day (Logan et al., 2006). These are bio-electrochemical devices which uses microbial culture for the electricity production. The microbial flora generates electrons, by utilizing various organic substrates, which is used for the bioelectricity productionas explained by (Oh et al., 2004). The substrates that have been reported to use as organic source in the microbial fuel cell are of many types. These substrates could be simple compounds like sucrose, glucose, fructose ethanol and acetate (Logan et al., 2007; Jafary et al., 2013) or it may also be complex media like food waste (Goud et al., 2011; Li et al., 2013a) and different waste water including domestic, agriculture industrial waste water etc. (Nimje et al., 2012).
Ionic strength is one of the important aspect due to which the internal resistance (Rint) in the microbial fuel cell is affected. Early studies done by Li et al. (2013b) showed that by increasing the internal resistance the power densities are decreased. In some other studies it has been suggested that increasing the NaCl concentration decrease the internal resistivity by improving the ionic strength and increase the energy production as explained by Kyung et al. (2004) and Lefebvre et al. (2012). Some studies conducted by Huang et al. (2008) also suggested that as the micro flora in the anodic chamber varies the salt tolerance may affect the bioelectricity production.
The bioelectricity production through MFC is greatly effect by the pH of the anodic chamber (He et al., 2008). It has been found, by changing the pH of the substrate in anodic chamber, that many physiological changes may occur like ionic strength, membrane potential, microbial metabolism and proton generation potential (Zhang et al., 2010; Jung et al., 2011). The ionic strength of the MFC can be improve by adjusting the pH of the anode chamber which can increase the power output by decreasing the internal resistance (Li et al., 2013b). In case of the dual chamber microbial fuel cell it has been found the higher pH difference in the two chambers can increase the power density (Jadhav and Ghangrekar, 2009). If alkaliphilic bacteria have been used the high substrate pH and low cathode pH has been found to give the maximum power output (Liu et al., 2010; Zhuang et al., 2010) while in some other studyit has been showed that lower substrate pH in anode give maximum potential (Rikame et al., 2012).
The substrate used in the MFC is one of the important factors which need to be optimized for higher power output. Food waste was also in interest of many researchers which serves as rich carbon and energy source for microorganism and also consists of almost 30% of the municipal waste. Huge variety of substrates have been used in the MFC as carbon sources for microbial culture such as glucose and fructose (Jafary et al., 2013), waste water (Nimje et al., 2012; Akman et al., 2013) and food waste (Goud et al., 2011; Li et al., 2013a). Molasses on the other hand not been extensively studied but some reports from past years showed molasses an effective electron donor in microbial fuel cells (Niessen et al., 2004; Fernando et al., 2014; Sevda et al., 2014).
MATERIALS AND METHODS
Substrate preparation
Vegetable waste from house hold kitchen was collected which included spinach leaves, cucumber and potato leaves, carrot and cabbage. These were converted into small pieces and blended with phosphate buffer solution (0.1 M) with pH 4.5 in 1:1 to solid weights of the waste pieces. The slurry was taken and filtered for further use.
Enrichment of microbial culture for inoculum preparation
Sewage sample was taken from the sewage waste water from Haal road, Lahore. Designed synthetic waste water (DSWW) was prepared as demonstrated by Venkata Mohan et al. (2010). The composition of DSWW is: K2HPO4-0.25g/l, KH2PO4-0.25g/l, NH4Cl-0.5g/l, ZnCl2-11.5mg/l, MgCl2-3g/l, CoCl2-25mg/l, CaCl2-5mg/l, CuCl2-10.5mg/l, MnCl2-15 mg/l, NiSO4-16mg/l, FeCl3-25mg/l, glucose-3g/land Vitamins-5g/l. The DSSW media was purged with nitrogen to provide anaerobic environment. The sample was inoculated in the DSWW media and incubated for 72 hours at 37°C. The enriched culture was centrifuged (6000 rpm at 20oC) and the pellet was washed thrice with saline phosphate buffer solution with 0.85% NaCl.
Microbial fuel cell configuration
Six U-shaped dual chambered MFCs with cation exchange membrane (CMI 7000) were prepared as explained in our previous study. All MFCs were supplemented with 200 ml substrate in anode but each with different NaCl concentration i.e. MFC-1 (25 mM), MFC-2 (50 mM), MFC-3 (75 mM), MFC-4 (100 mM), MFC-5 (125 mM) and MFC-6 (150 mM). Tap water was used as catholyte which already contains enough salts and traces for the process to run. For optimization of pH studies, six different MFCs were set at pH: 3.5, 4.5, 5.5, 6, 7 and 7.5. NaCl concentration was kept 50mM in all MFCs. Graphite rods were used as anode and cathode, three in each chamber, with total surface area of 3×0.0015 m2. The electrodes were connected to a multimeter (UNI-T UC- 60A), with personal computer (PC) interface, parallel to variable resistance (5-5000Ω) as shown in Figure 1.
Experimental design for utilization of molasses as substrate for bioelectricity production
Clarification of cane-molasses was performed by using the method of Panda et al. (1984). MFC-1 (10% sugar content with 9119mg/l COD), MFC-2 (15% sugar with 12076mg/l COD) and MFC-3 (20% sugar content with 15390mg/l COD) were set up for this experiment.NaCl (50mM) was added to provide sufficient ionic strength to media in all three MFCs. Cathode of MFC was supplemented with tap water (pH 7.0) whereas clarified molasses (pH 4.5) was added in anode chamber of MFC, along with mix microbial cultureenriched in synthetic waste water media as inoculum (Mohan et al., 2010).
Data analysis
Data was recorded across different resistance from 5-5000 Ω and the current was calculated by relation V/R where V is voltage and R is respective resistance and power was calculated using the formula P=VI, where I represents current. The power density and current density were calculated by dividing the electrode (anode) surface area to power and current, respectively. The slope of the linear zone of the graph between the current and voltage gives the internal resistance (Logan et al., 2006). Columbic Efficiency (C.E) was also calculated by checking the COD difference after specific time period. Following relation was used for the C.E in which ΔCOD is change in chemical oxygen demand, I is current, V is substrate volume in anode, b is total number of electrons i.e. 4 per mol of oxygen, F is faraday’s constant, and M is molar weight of oxygen.
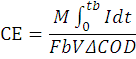
RESULTS AND DISCUSSION
Performance of MFC at different NaCl concentration
The result from two ranges of the salt treatments, i.e. lower range (25 to 75 mM) and higher range i.e. (100 to 150 mM), showed that with higher salt concentration the microbial fuel cell performance decreased which is the evidence of lower microbial activity and increased internal resistivity (Fig. 2) due to volumetric resistance as described by Clauwaert et al. (2008). Li et al. (2013b) also suggested that NaCl concentration improves the ionic strength which helps in electron and proton transfer through the microbial fuel cell. The internal resistance of the higher range treatments is steady but with slight changes with time as compare to lower treatments which showed to be the reason of higher microbial activity in the lower range NaCl concentration and vice versa which could be the optimal requirement for microorganism as explained by Prescott (2002). The higher microbial activity also leads the MFC to increase the power density which is why higher power output was noted at lower range of salt concentration (Fig. 2). The results confirms the optimum concentration of salt should be needed for efficient production of bioelectricity and MFC performance which was also previously studied by Li et al. (2013a) which might be different for a MFC depending on the substrate and microbial culture used.
The current was calculated by observing the voltage on the external load as described by Logan et al. (2006). It was found that 200Ω of external resistance provides maximum power density which was to be selected for analysis. The maximum power production was 88.99 W/m2 at 50 mM NaCl concentration on the second day of the process while 67.78 W/m2 and 81.12 W/m2 power densities was for the first and third day, respectively (Fig. 2). The power output from this work is higher, to our knowledge, than most of the reports by Wang et al. (2014); Yong et al. (2014); Cao et al. (2015). The concentration of 50 mM then other concentration confirms the optimized condition for running the MFC for this work. Although other lower ranged concentration was also quite significant and closer each other in term of the open circuit voltage and power density (Figs. 1 and 2). The lower range support higher power output because of efficient microbial activity and lower internal resistance as compare to the higher range of the NaCl concentration as described by Li et al. (2013b).
Effect of pH on power generation in MFC
By keeping the cathodic chamber pH constant, i.e. 7 to 7.5, the anodic chamber pH is being decreased in which the increase in the power generation occur gradual decrease in the pH up to a certain limit. Changes in the pH may result various physiological changes in the Microbial fuel cell like changes in membrane potential, microbial metabolism and ionic strength especially (Zhang et al., 2010; Jung et al., 2011). The increase in power with decrease in pH may due to the creation of pH gradient between the anodic and cathodic chamber which resulted the flow of ionic movement from one to another chamber efficiently (Jadhav and Ghangrekar, 2009; Zhuang et al., 2010). Although the result was in contradiction with the reports of Zhuang et al. (2010) whose power output was higher at higher anodic and lower cathodic pH but in this case higher potential was at pH 4.5 with power density of 70.5 W/m2 (Fig. 3) which was higher than reported by Zhuang et al. (2010) but when the pH is decreased to 3.5 the power and voltage generation was getting low (Fig. 3) which indicated the decrease in the microbial activity with respect to the conditions (Gaboriaud et al., 2006). In some other reports the acidic pH of anode chamber gave higher power output (Rikame et al., 2012; Li et al., 2013b).
The MFCs with lower range pH i.e. MFC-1, 2 and 3 with pH 3.5, 4.5 and 5.5, respectively gave maximum power output and open circuit voltage (Fig. 3) due to higher pH difference between cathodic and anodic chamber (Zhang et al., 2010; Rikame et al., 2012; Li et al., 2013b). The higher power density was 70.5 W/m2 observed at the first day of the process in the MFC-2 with pH 4.5 (Fig. 3). While the second and third day was also higher than the rest of the MFCs which was almost 51 W/m2 except to the first day process of MFC-1 which was 69.3 W/m2. The result was higher than most of the other reports to our knowledge (Li et al., 2013b; Wang et al., 2014; Yong et al., 2014; Cao et al., 2015). As the pH gets closer to the cathodic pH which was 7 to 7.5 the lower power output was observed. It also has been observed that at pH 6 and 7.5 the internal resistance was minimum (Fig. 3) which is assumed to be the reason of optimized condition for ionic strength for minimum internal resistance with respect to the cathodic chamber (Li et al., 2013b). The results showed that MFCs with lower ranged pH gave higher potential but maximum ohmic losses as compare to those with higher pH treatments with one unit difference as referred to cathode pH (Clauwaert et al., 2008).
Efficiency of microbial fuel cell and electricity generation with molasses as substrate
It has been found that the C.E of MFC-2 (81.7%) was higher, compared to MFC-1 (73.9%) and MFC-3 (63.2%). Lower efficiency of the MFC-1 and MFC-3 can be reasoned due to lower microbial metabolic rate, for which higher internal resistance comparatively to the current production might reduced the coulombic efficiency. Same pattern of results has been reported by Rikame et al. (2012) and Juang et al. (2011).
Voltage was analyzed on variable external load and a straight line was observed up to 400Ω for MFC-1 and MFC-3 while in case of MFC-2, straight line goes to 500 Ω, which shows that higher power density can be obtained at these external loads (Fig. 4). MFC-1 recorded maximum power density (72820mW/m2, at 400Ω) as compared to MFC-3 (44340mW/m2, at 500Ω) and MFC-2 (104400mW/m2, at 500Ω) (Fig. 5). Internal resistance of MFC-2 was higher as compare to other two i.e. 283.3Ω (Fig. 6) but higher power and electric-current output is the evidence of the high activity of microorganisms comparatively. The results of power density in present study was higher than most of the reports using the molasses as electron donor (Niessen et al., 2004; Fernando et al., 2014; Sevda et al., 2014).
Kargi and Eker (2007) used molasses with urea and reported power density of 2.6mW/m2. While in comparison to other substrates, molasses was found to be an effective substrate in term of power production. Mansoorian et al. (2013) reported power density of 230mW/m2 from food processing waste water. Whereas, relatively higher power densities of 590mW/m2 (xylose as substrate) and 1500mW/m2 (chocolate industry wastewater as substrate) has been reported by Mäkinen et al. (2013) and Patil et al. (2009), respectively.
Results of present work also favor open circuit voltage of MFC-2 with average value of 471mV while MFC-1 and MFC-3 had 338mV and 312mV of potential difference, respectively (Fig. 7). Open circuit voltage recorded in present work is lower than some reports in which different kinds of substrate have been used (Zhuang et al., 2010; Rikame et al., 2012; Wang et al., 2014). This could be possibly explained by the existence of higher volumetric resistivity in cane molasses as compared to other substrates (Clauwaert et al., 2008).
CONCLUSION
Sodium chloride is common salt that is been used in homes gave us most economic choice to use as ionic agent for increasing the ionic strength in the Microbial Fuel Cell. The optimum concentration of the salt can increase the power density by minimizing the internal resistance. Increasing the salt concentration from the optimum level might result in lower power output due to increased internal resistance and lower microbial metabolic rate to produce electrons. It has been found that pH is a crucial element in the optimizing and increasing the power of the microbial fuel cell. If the microbial fuel cell is dual chamber it should be necessary to increase the pH difference up to a limit between the two chambers for ionic facilitation and higher voltage production. The choice of microorganism should be more like acidophilic for low anode pH or alklaphilic for high anode pH, to support other findings.
Present study also revealed that molasses can act as good initiating agent or carbon source for microorganism in microbial fuel cells for bioelectricity production. Literature reports suggested the use of other substrates for bioelectricity production but problem of lower internal resistivity with these substrates empowers the fact of molasses as potent substrate in MFCs.
ACKNOWLEDGEMENT
We are highly grateful to Higher Education Commission of Pakistan for facilitating us in the research work.
Statement of conflict of interest
The authors have declared no conflict of interest.
REFERENCES
Akman, D., Cirik, K., Ozdemir, S., Ozkaya, B. and Cinar, O., 2013. Bioelectricity generation in continuously-fed microbial fuel cell: Effects of anode electrode material and hydraulic retention time. Bioresour. Technol.,149: 459-464. https://doi.org/10.1016/j.biortech.2013.09.102
Cao, X., Song, H., Yu, C. and Li, X., 2015. Bioresource technology simultaneous degradation of toxic refractory organic pesticide and bioelectricity generation using a soil microbial fuel cell. Bioresour. Technol., 189: 87-93. https://doi.org/10.1016/j.biortech.2015.03.148
Clauwaert, P., Aelterman, P., Pham, T.H., De Schamphelaire, L., Carballa, M., Rabaey, K. and Verstraete, W., 2008. Minimizing losses in bio-electrochemical systems: The road to applications. Appl. Microbiol. Biotechnol., 79: 901-913. https://doi.org/10.1007/s00253-008-1522-2
Fernando, E., Keshavarz, T. and Kyazze, G., 2014. Bioresource technology complete degradation of the azo dye Acid Orange-7 and bioelectricity generation in an integrated microbial fuel cell, aerobic two-stage bioreactor system in continuous flow mode at ambient temperature. Bioresour. Technol., 156: 155. https://doi.org/10.1016/j.biortech.2014.01.036
Gaboriaud, F., Dague, E., Bailet, S., Jorand, F., Duval, J. and Thomas, F., 2006. Multiscale dynamics of the cell envelope of Shewanella putrefaciens as a response to pH change. Colloid Surf. B: Biointerf., 52: 108-116. https://doi.org/10.1016/j.colsurfb.2006.04.017
Goud, R.K., Babu, P.S. and Mohan, S.V., 2011. Canteen based composite food waste as potential anodic fuel for bioelectricity generation in single chambered microbial fuel cell (MFC): Bio-electrochemical evaluation under increasing substrate loading condition. Int. J. Hydrog. Energy, 36: 6210-6218. https://doi.org/10.1016/j.ijhydene.2011.02.056
He, Z., Huang, Y., Manohar, A.K. and Mansfeld, F., 2008. Effect of electrolyte pH on the rate of the anodic and cathodic reactions in an air-cathode microbial fuel cell. Bioelectrochemistry, 74: 78-82. https://doi.org/10.1016/j.bioelechem.2008.07.007
Huang, L., Zeng, R.J. and Angelidaki, I., 2008. Electricity production from xylose using a mediator-less microbial fuel cell. Bioresour. Technol., 99: 4178-4184. https://doi.org/10.1016/j.biortech.2007.08.067
Jadhav, G.S. and Ghangrekar, M.M., 2009. Performance of microbial fuel cell subjected to variation in pH, temperature, external load and substrate concentration. Bioresour. Technol., 100: 717-723. https://doi.org/10.1016/j.biortech.2008.07.041
Jafary, T., Rahimnejad, M., Ghoreyshi, A.A., Najafpour, G., Hghparast, F. and Daud, W.R.W., 2013. Assessment of bioelectricity production in microbial fuel cells through series and parallel connections. Energy Convers. Manage., 75: 256-262. https://doi.org/10.1016/j.enconman.2013.06.032
Jung, S., Mench, M.M. and Regan, J.M., 2011. Impedance characteristics and polarization behavior of a microbial fuel cell in response to short-term changes in medium pH. Environ. Sci. Technol., 45: 9069-9074. https://doi.org/10.1021/es201737g
Kargi, F. and Eker, S., 2007. Electricity generation with simultaneous wastewater treatment by a microbial fuel cell (MFC) with Cu and Cu-Au electrodes, J. Chem. Tech. Biotechnol., 82: 658. https://doi.org/10.1002/jctb.1723
Kyung, J., Hai, T., Seop, I., Hyun, K., Moon, H., Suk, K. and Hong, B., 2004. Construction and operation of a novel mediator- and membrane-less microbial fuel cell. Process Biochem., 39: 1007-1012. https://doi.org/10.1016/S0032-9592(03)00203-6
Lefebvre, O., Tan, Z., Kharkwal, S. and Ng, H.Y., 2012. Effect of increasing anodic NaCl concentration on microbial fuel cell performance. Bioresour. Technol., 112: 336-340. https://doi.org/10.1016/j.biortech.2012.02.048
Li, X.M., Cheng, K.Y. and Wong, J.W.C., 2013b. Bioelectricity production from food waste leachate using microbial fuel cells: Effect of NaCl and pH. Bioresour. Technol., 149: 452-458. https://doi.org/10.1016/j.biortech.2013.09.037
Li, X.M., Cheng, K.Y., Selvam, A. And Wong, J.W.C., 2013a. Bioelectricity production from acidic food waste leachate using microbial fuel cells: Effect of microbial inocula. Process Biochem., 48: 283-288. https://doi.org/10.1016/j.procbio.2012.10.001
Liu, M., Yuan, Y., Zhang, L.X., Zhuang, L., Zhou, S.G. and NI, J.R., 2010. Bioelectricity generation by a Gram-positive Corynebacterium sp. strain MFC03 under alkaline condition in microbial fuel cells. Bioresour. Technol., 101: 1807-1811. https://doi.org/10.1016/j.biortech.2009.10.003
Logan, B., Cheng, S., Watson, V. and Estadt, G., 2007. Graphite fiber brush anodes for increased power production in air-cathode microbial fuel cells. Environ. Sci. Technol., 41: 3341-3346. https://doi.org/10.1021/es062644y
Logan, B.E., Hamelers, B., Rozendal, R., Schröder, U., Keller, J., Freguia, S., Aelterman, P., Verstraete, W. And Rabaey, K., 2006. Microbial fuel cells: Methodology and technology. Environ. Sci. Technol., 40: 5181-5192. https://doi.org/10.1021/es0605016
Mäkinen, A.E., Lay, C.H., Nissilä, M.E. and Puhakka, J.A., 2013. Bioelectricity production on xylose with a compost enrichment culture. Int. J. Hydrog. Energy., 38: 15606. https://doi.org/10.1016/j.ijhydene.2013.04.137
Mansoorian, H.J., Mahvi, A.H., Jafari, A.J., Amin, M.M., Rajabizadeh, A. and Khanjani, N., 2013. Bioelectricity generation using two chamber microbial fuel cell treating wastewater from food processing. Enzyme. Microb. Technol., 52: 352. https://doi.org/10.1016/j.enzmictec.2013.03.004
Mohan, S.V., Mohanakrishna, G. and Sarma, P.N., 2010. Composite vegetable waste as renewable resource for bioelectricity generation through non-catalyzed open-air cathode microbial fuel cell. Bioresour. Technol., 101: 970. https://doi.org/10.1016/j.biortech.2009.09.005
Niessen, J., Schröder, U. and Scholz, F., 2004. Exploiting complex carbohydrates for microbial electricity generation-A bacterial fuel cell operating on starch. Electrochem. Commun., 6: 955. https://doi.org/10.1016/j.elecom.2004.07.010
Nimje, V.R., Chen, C.Y., Chen, H.R., Chen, C.C., Huang, Y.M., Tseng, M.J., Cheng, K.C. and Chang, Y.F., 2012. Comparative bioelectricity production from various wastewaters in microbial fuel cells using mixed cultures and a pure strain of Shewanella oneidensis. Bioresour. Technol., 104: 315-323. https://doi.org/10.1016/j.biortech.2011.09.129
Oh, S., Min, B. and Logan, B.E., 2004. Cathode performance as a factor in electricity generation in microbial fuel cells. Environ. Sci. Technol., 38: 4900-4904. https://doi.org/10.1021/es049422p
Panda, T., Kundu, S. and Majumdar, S.K., 1984. Studies on citric acid production by Aspergillus niger using treated Indian cane molasses. J. Microbiol., 52: 61.
Patil, S.A., Surakasi, V.P., Koul, S., Ijmulwar, S., Vivek, A., Shouche, Y.S. and Kapadnis, B.P., 2009. Electricity generation using chocolate industry wastewater and its treatment in activated sludge based microbial fuel cell and analysis of developed microbial community in the anode chamber. Biores. Technol., 100: 5132. https://doi.org/10.1016/j.biortech.2009.05.041
Prescott, L.M., Harley, J.P. and Klein, D.A., 2002. Microbiology 5th Edition. The McGraw-Hill Companies, New York City, USA.
Rikame, S.S., Mungray, A. and Mungray, A.K., 2012. Electricity generation from acidogenic food waste leachate using dual chamber mediator less microbial fuel cell. Int. Biodeterior. Biodegrad., 75: 131-137. https://doi.org/10.1016/j.ibiod.2012.09.006
Sevda, S., Dominguez-Benetton, X. and Wever, H.D., 2014. Evaluation and enhanced operational performance of microbial fuel cells under alternating anodic open circuit and closed circuit modes with different substrates. Biochem. Eng. J., 90: 294. https://doi.org/10.1016/j.bej.2014.06.024
Venkata Mohan, S., Mohanakrishna, G. and Sarma, P.N., 2010. Composite vegetable waste as renewable resource for bioelectricity generation through non-catalyzed open-air cathode microbial fuel cell. Bioresour. Technol., 101: 970-976. https://doi.org/10.1016/j.biortech.2009.09.005
Wang, J., Zheng, Y., Jia, H. and Zhang, H., 2014. Bioresource technology bioelectricity generation in an integrated system combining microbial fuel cell and tubular membrane reactor: Effects of operation parameters performing a microbial fuel cell-based biosensor for tubular membrane bioreactor. Bioresour. Technol., 170: 483-490. https://doi.org/10.1016/j.biortech.2014.08.033
Yong, X., Feng, J., Chen, Y., Shi, D., Xu, Y., Zhou, J., Wang, S., Xu, L., Yong, Y., Sun, Y., Shi, C., Ouyang, P. and Zheng, T., 2014. Biosensors and bioelectronics: enhancement of bioelectricity generation by cofactor manipulation in microbial fuel cell. Biosens. Bioelectron., 56: 19-25. https://doi.org/10.1016/j.bios.2013.12.058
Zhang, X., Cheng, S., Huang, X. and Logan, B.E., 2010. Improved performance of single-chamber microbial fuel cells through control of membrane deformation. Biosens. Bioelectron., 25: 1825-1828. https://doi.org/10.1016/j.bios.2009.11.018
Zhuang, L., Zhou, S., Li, Y. and Yuan, Y., 2010. Enhanced performance of air-cathode two-chamber microbial fuel cells with high-pH anode and low-pH cathode. Bioresour. Technol., 101: 3514-3519. https://doi.org/10.1016/j.biortech.2009.12.105
To share on other social networks, click on any share button. What are these?










