Effects of a Mixture of Acacia Honey and Povidone-Iodine on the Healing of Full Thickness Skin Wound in Normal and Dexamethasone Treated Rabbits
Effects of a Mixture of Acacia Honey and Povidone-Iodine on the Healing of Full Thickness Skin Wound in Normal and Dexamethasone Treated Rabbits
Muhammad Awais1,2, Waqas Ahmad3, Akhtar Rasool Asif4, Sayyed Aun Muhammad1, Iahtasham Khan1*, Muhammad Amjad Ali5, Muhammad Qaiser Riaz1 and Muhammad Saqib2
1Department of Clinical Sciences, College of Veterinary and Animal Sciences, Jhang, Pakistan
2Department of Clinical Medicine and Surgery, Faculty of Veterinary Sciences,
University of Agriculture, Faisalabad, Pakistan
3Department of Clinical Sciences, KBCMA University College of Veterinary and Animal Sciences, Narowal, Pakistan
4Department of Animal Sciences, College of Veterinary and Animal Sciences, Jhang,
Pakistan
5Department of Clinical Sciences, Faculty of Veterinary Sciences, Bahauddin Zakariya University, Multan, Pakistan
ABSTRACT
Honey is considered as the oldest known traditional medicine being used for a variety of illnesses and healing of wounds. A total of 40 rabbits were divided into four groups (A, B, C, and D) to perform two experiments. Two wounds of full-thickness with the dimensions of 1×1 cm (1 cm2), were created on right and left lateral sides of the trunk, comprising 4 wounds on each rabbit. Wounds of group A, B, C and D were treated with pyrogen free normal saline, 0.05% PI gel, certified Acacia honey and H-PImxr (an equal volume mixture of Acacia honey + Pyodine gel) respectively. Significant differences were found among four treatments with respect to contraction, granulation tissue, tensile strength, healing time and hydroxyproline contents measurement in both experiments. Honey and povidone mixture treated animals showed significantly improved wound contraction, healing time, tensile strength and hydroxyproline measurements in dexamethasone treated group. In normal group, honey treated animals have significantly reduced wound healing time than the honey and povidone treated animals in dexamethasone treated animals. In normal group, honey and povidone treated animals exhibited significantly improved results for wound contraction, tensile strength as compared to animals treated with honey alone. Honey in combination with povidone enhanced wound healing by improving wound contraction rate, tensile strength, hydroxyproline contents and by reducing wound healing time. The use of honey in combination with povidone iodine instead of using either honey or povidone alone for future treatment of wounds in normal and dexamethasone treated animals is suggested.
Article Information
Received 02 February 2019
Revised 21 September 2019
Accepted 01 October 2019
Available online 21 April 2021
(early access)
Published 17 December 2021
Authors’ Contribution
MA, MS designed the study plan and performed experiments. AM and ARA induced surgical wounds and prepared mixtures. MQR maintained experimental animals and performed statistical analysis. MAA and IK conceived the study plan and wrote manuscript. WA analyzed the data and helped in preparation of the manuscript.
Key words
Wound healing, Acacia honey, Povidine-iodine, Dexamethasone, Rabbits
DOI: https://dx.doi.org/10.17582/journal.pjz/20190202100216
* Corresponding author: Iahtasham.khan@uvas.edu.pk
0030-9923/2022/0001-0391 $ 9.00/0
Copyright 2022 Zoological Society of Pakistan
INTRODUCTION
Delayed wound-healing poses a serious challenge in clinical practice (Hess, 2011). About 3-6 million people are affected by non-healing wounds in United States, among them 85% are 65 years age or above. Corticosteroids are widely used in clinical practice to treat severe inflammatory reactions and immune-mediated disorders. The adverse effects of steroids on wound healing (impaired/delayed healing) are well known since their discovery (Levine, 2017). Steroids inhibit inflammatory phase (decrease vascular permeability, extravasation, inhibit complement and lymphocyte derived factors), angiogenesis, fibroblastic proliferation, and extracellular matrix (proteins and proteoglycans) synthesis (Horng et al., 2017; Levine, 2017). In general steroids delay epithelization, wound strength and closure (Chamanga, 2018). It has been found that steroids inhibit both epithelization and contraction at any stage of wound healing (Swaim et al., 2001). Often time surgeries are performed on patients already suffering from immune-mediated disorders or receiving steroidal therapies for disease management purposes. In such instances, post surgical wound care is important to avoid extra-delay in healing process. Simple and effective wound healing remedies are warranted to investigate their role in animal models before any application in clinical practice.
Honey, a natural food produced by honey bee (Apis mellifera) is the oldest known traditional medicine being used for a variety of illnesses and healing of wounds (Tan et al., 2012). Several studies have documented a salubrious healing effect of honey on different types of wounds. Honey has a barrier effect on open wound, which prevent the wound from external contamination (Molan, 2011). Among four Pakistani honeys, Acacia honey has been found superior to Ziziphus, Brasicca and Citrus honeys in terms of antimicrobial activity, total sugar and protein contents (Gulfraz et al., 2011).
Povidone-iodine (PVP-I; PI), a commonly used iodophor is chemical complex of polyvinylpyrrolidone (PVP) and element iodine (I). Over the last 35 years, around 27 randomized clinical trials have documented effectiveness of iodine and iodine containing compounds in wound infection and care (Bigliardi et al., 2017). Cadexomer iodine (polysaccharide beads containing 0.9% iodine) accelerates healing through minimizing the bio-burden of wounds, removing necrotic tissue and stimulating epidermal regeneration (Daniel et al., 2016; Johani, 2017). Mixture of PI (3%) and saturated sugar solution (70%) accelerates healing of chronic wounds (e.g., burns and diabetic ulcers) through reducing bio-burden, influencing migration of cells (keratinocytes and fibroblast) and stimulating granulation tissue formation (Shi et al., 2007). This mixture is commonly used in Japan for treatment of diabetic and pressure ulcers (Shi et al., 2007). In view of the foregoing, a preliminary study was planned to compare the healing effect(s) of mixture of honey-povidone iodine (H-PImxr) with PI (a standard wound management protocol) on normal and dexamethasone suppressed full thickness skin wounds in rabbits. It was hypothesized that (H-PImxr) would have providing a rapid healing (promoting fibroplasias, contraction and closure). A null hypothesis formulated was that there would not be differences in appearance of granulation tissue, fibroplasias and contraction rate between the treatments.
MATERIALS AND METHODS
The present hypothesis was tested on adult (n=40) rabbits in normal and dexamethasone treated phases, making a total of one hundred and sixty surgically inflicted full thickness skin wounds on the torso (trunk; thoraco-lumber region). The study was ethically approved from the ethical scrutiny committee of the University of Agriculture Faisalabad, Pakistan.
The locally bred, clinically healthy male adult rabbits (n=20), weighing about 2.5-3kg were purchased from single vender and randomly divided into 4 groups, viz, A, B, C and D. All the rabbits were maintained on a uniform feeding and management regime during both pre-and post-operative periods.
Before experimentation, all animals were acclimatized for two weeks in the Laboratory Animal Facility of the Department of Clinical Medicine and Surgery with 8-10 h light-14-16 h dark cycle at 22-28°C room. During this period, each rabbit was administered with 02 subcutaneous doses of ivermectin (400 µg/kg) a week apart and a 3-day prophylactic course of amoxicillin (15mg/kg) for pasturellosis that terminated a week before commencement of experimental trial. Animals were fed on green fodder (berseem) twice a day and dry pellet feed once in day. Water was offered generally, ad libitum. The operation site was prepared for aseptic surgery by spraying methylated spirit twice and tincture of iodine once after the other, after clipping of hair on the torsa.
Each rabbit was pre-medicated by administering atropine sulphate 0.035mg/kg body weight through s/c route half an hour prior to surgical intervention. Animals were anesthetized by total parenteral (intramuscular) anesthesia using a mixture of ketamine (35mg/kg) and xylazine (5mg/kg) (Lipman et al., 1990).
Wounding and application of treatments
Wounding area was marked with permanent maker using X-Rays’ wound template. Two, 1×1 cm (or 1cm2) full-thickness skin wound was created bilaterally on the trunk comprising 4 wounds on each rabbit. Each wound was 2cm away from dorsal mid line and 4 cm apart on each side of the spine. Full thickness wound was created on the trunk with the help of #11 blade (Carbon Steel, Feather Safety Razor Co, Ltd. Japan) and the piece of skin from wound was removed by undermining the area with a sharp-sharp scissors leaving subcutaneous area exposed for further studies (Supplementary Fig. 1 to 4). Another similar wound was created on the area adjacent to the first wound.
Four wounds of similar dimensions were created on the dorsum of each rabbit. The wounds of group A designated as follows for the proper identification and avoid repetition of treatment: A-wRF, wound created lateral to the thoraco-lumber area on right front of the body; A-wRR, wound created lateral to the thoraco-lumber area on right rear of the body; A-wLF, wound created lateral to the thoraco-lumber area on left front of the body; A-wLR, wound created lateral to the thoraco-lumber area on left rear of the body. A similar naming was assigned to the wound on subject of the groups B, C and D.
Wounds in group A received application of certified Acacia honey (0.5 ml/wound) obtained from NARC Islamabad, Pakistan right from the day of operation. Similarly, wounds in groups B were treated with a mixture of Acacia honey and povidone iodine (200g of povidine iodine gel mixed in equal amount with acacia honey). The subjects in groups C received treatment with povidone iodine gel (Pyodine gel, 0.05% Brookes Pharmaceutical Lab, Pakistan) and rabbits of groups D served as control and received pyrogen free normal saline wash as a treatment.
After application of each treatment, the wounds were covered with gamma sterilized cotton gauze (Steriband gaze, Faisal Pharmaceutical Industries, Pak) and properly secured with cotton bandage. The dressings were changed on daily basis until complete healing.
Dexamethasone treatment
Another group of 20 rabbits as described above received dexamethasone (@2mg/kg body weight, 6 hourly) intramuscularly a day before wounding (6 hourly interval) and during treatment period (@2mg/kg, 48 hourly) till complete healing (Jeklova et al., 2008).
Evaluation parameters for wound healing
The wound healing in all treatment groups was estimated by undertaking the following healing parameters:
Wound contraction rate
Wound contraction rate was estimated by tracing the edges of wounds on piece of trace paper at 3 days interval until complete healing/epitheilization. These sketches were measured by graph paper (mm2) and wound contraction values were presented in percentile.
Healing time
Healing time was estimated by sum of daily observations till scar dropped off.
Tensile strength
On complete healing, tensile strength of regenerated tissue was measured especially devised for measuring the tensile strength of fabrics, vinyl, rubber, copper wire by tensile testing machine (Tensometer, Japan) available in the Department of Fiber Technology. The stripes of 1cm wide and 20 cm long was prepared by scalpel blade from all treated and control groups and were loaded to the machine. This load value (force) was recorded. The elongation of the specimen was continued until a rupture of the specimen was observed. The breaking strength was calculated (Athar et al., 1996) using following formulae:
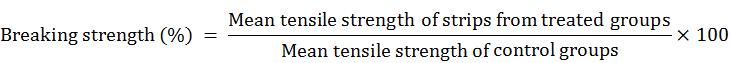
Estimation of hydroxyproline contents in healed tissues
On complete healing, contents of hydroxyproline were determined in healed tissues by using a commercially available hydroxyproline determination kit (QuickZyme Hydroxyproline Assay, QuickZyme BioSciences, Netherlands).
Statistical analysis
The values of parameters i.e., healing time, contraction rate, and tensile strength and hydroxyproline contents were presented as mean ± SE. Significance among different treatments was determined by ANOVA using statistic calculator’s version 3. The values were considered significant at P <0.05.
RESULTS
Wound contraction rate
Figure 1 shows wound contraction rate in the untreated rabbits. The contraction subsided by day 21 in wounds treated with honey while contraction phase terminated on day 25, in groups B, C and D. The highest wound contraction rate (3.38mm = 33.8%) was registered in honey treated group on day 5. At this time point, percent contraction rate was 22.9 (2.29mm), 21.7 (2.17mm) and 21 (2.1mm), respectively for NS, PI, and H-PImxr treated groups (Fig. 1). Statistically the contraction rate was significantly higher (p=0.001) in honey treated group than other treatments. On comparing the statistics between honeys versus other treatment groups (PI; H-PImxr, NS) on wound contraction rate by day 5, highly significant differences (p=0.003 for H vs. PI and H-PImxr; p = 0.007 for H vs NS) were noted. By day 15, the results of influence of different treatments on contraction rate (means of four wounds of each rabbit) were noted insignificant. By day 15, the contraction rates of H vs H-PImxr and H vs PI were found insignificant (P=0.376 and p= 0.096), however, rate contraction was significant (p <0.035) faster in honey treated group vs untreated control (NS).
Among dexamethasone treated groups, on day 5, highest reduction (29.6. % = 2.96 mm) in wound size was noted in H-PImxrtreated animals (group B), followed by 21.27 %, 16.7 %, respectively in PI (group C) and honey (group A) treated groups. On this time point (day 5) a markedly low contraction rate (15 %= 1.5mm) was registered in normal saline treated group (Fig. 2). On day 15thof treatment, wound edges covered 50% (5mm) of the wounds area in groups B (H-PImxr) C (PI). However, al low contraction rate (34.9-35.5%) was observed in honey and normal saline treated groups, statistically differences were non-significant (p = 0.883). The contraction rate was highly significantly (p = 0.00) in wounds treated with H-PImxr than wound in group A (honey) and C (pyodine) alone. On comparing the effect of H-PImxr on contraction rate of untreated versus treated wounds, again significantly higher (p=0.016) wound contraction was observed (Supplementary Figures 1 to 5).
Healing time
In untreated rabbits, the lowest mean wound healing time (19.35days ±1.633) was noted in groups A (treated with honey), followed by group C (povidone iodine; 21.52.11), group D (21.8±1.163) and group B (22.3±0.41). Healing was statistically significantly faster (P< 0.036) in honey treated wounds than that of other the treatments. Non-significant differences were noted among PI, H-PImxr and NS treatments.
Among dexamethasone treated groups, the lowest average wound healing time (20.4 days± 3.89) was noted with mixture of honey-povidone iodine followed by treatment with iodine (28.25 ± 1.65) and honey (30.2±1.40). Wounds treated with normal saline (control or group D) healed on day 31. Healing time was significantly lower (0.001) in H-PImxr treated wounds than treatments with honey and PI alone. Differences between treatment with honey and PI were statistically non-significant (p<0.079). A significant difference (p<0.06) was noted between povidone iodine verses normal saline healing time.
Tensile strength
Tensile strength (TS) was statistically significantly high (p<0.002 and p<0.001) with honey-povidone iodine treated wounds in both experiments than povidone iodine and honey alone. Differences between TS of treatments of two experiments (honey vs honey, PI vs PI, H-PImxrvs H-PImxr) were non-significant (p<0.063; 0.088 and 0.167).
Hydroxyproline contents
Hydroxyproline contents of dexamethasone (DXM) healed wounds were significantly lower (0.001) than normal wounds, however, statistically higher values were noted in H-PImxr treatments in both DXM treated and normal wounds.
DISCUSSION
In the present study, no significant (p>0.05) difference was found in the entire physiological parameter viz. temperature and respiration rates. A slight rise in temperature, plus and respiration rates were recorded on day one postoperative. In both groups which returned to normal on day five onward. A slight rise in temperature was because of body reaction to the surgical trauma and possibly preoperative mild contamination (Bindu et al., 2017). Present study results are in agreement with those of Ather et al. (1996) who considered the change in temperature as a normal stress responsible to the surgical insult. Tensile strength was better in honey alone and in combination with pyodineiodine which might be due to the Cu and Zn contents of the honey. Cu induces solid and stable collagen at the site of wound. Zn and Cu on the other hand act as a cofactor of superoxide dismutase enzyme which prevents the body from oxygen radicals. Zn acts as a cofactor of collagenase enzyme which is further involved in the collagen synthesis (Lin et al., 2018; Marreiro and Kyria, 2017; Kornblatt et al., 2016), so in this way Zn is important for wound healing with respect to tensile strength and this can be correlated with the results of our study which showed better tensile strength in honey alone and in combination with pyodine iodine treated rabbits. Additionally, increased tensile strength in honey alone and honey plus pyodine iodine might be due to increased density, keratinization, granulation tissue and activation of fibroblasts thus encouraging the collagen synthesizing surface of wound (Arai et al., 2013). Development of granulation tissue and wound contraction is related to each other in that granulation tissue contains myofibrils, which are responsible for the wound contraction. In the present study, wound contraction rate were quick in honey along with povidone treated animal in dexamethasone treated group then other groups (treated with either alone honey, alone povidone, or alone normal saline). In other normal group the wound contraction rate were quick in alone honey treated group then other with combination of povidone, alone povidone and normal saline treated animal (Benjamin and Bowlin, 2018; Eyarefe et al., 2017).
In present study, wound healing time was reduced for honey along with pyodine iodine treated rabbits. This might be due to the sugars present in honey which in combination with pyodine iodine enhanced the formation of granulation tissue, number of capillaries and re-epithelialization (Bobis et al., 2018; Benjamin and Bowlin, 2018). The reason for improvement in wound healing with honey alone is still unclear so far but probably it might be due to the antibacterial and anti-inflammatory properties. Honey induces wound healing by reducing ROS levels, lowering the pH and high free acid content which discourage the growth of microbes (Ahmed et al., 2018; Samarghandian et al., 2017). Some additional favorable properties of wound are its easy availability, formations of physical barrier against microbes. Honey creates a moist environment at the site of wound hence encouraging the process of healing. Honey decreases inflammatory edema, hastens sloughing of devitalized tissue, attracts macrophages which cleanse the wound, provides a local cellular energy source, and protectively covers the wound. (Almasaudi et al., 2015) A high osmolarity, acidity, and hydrogen peroxide content confer honey with antibacterial properties. Honey also appears to be more effective than sugar in reducing bacterial contamination and promoting wound healing and slightly less painful than sugar during dressing changes and motion (Molan and Rhodes, 2015). Honey debrided wounds rapidly, replacing sloughs with granulation tissue (Bobis et al., 2018; Ahmed et al., 2018; Samarghandian et al., 2017). It also promoted rapid epithelialization, and absorption of edema from around the ulcer margins. However, there is a tendency for some practitioners to dismiss out of hand any suggestion that treatment with honey is worthy of consideration as a remedy in modern medicine. Wound strength is determined by the amount and quality of newly synthesized and deposited collagen, as well as degradation of preformed collagen. The net amount of wound collagen deposition depends on collagen turnover and is a reflection of collagen synthesis minus collagen breakdown. The changes in the diameter of collagen fibrils have also been related to mechanical strength of the skin. Apparently thick collagen fibrils can resist greater tensile strength as opposed to thin ones. Once the skin is injured, the normal collagen will be replaced by scar collagen and the connective tissue will not regain the original highly organized structure of collagen.
There is supplementary material associated with this article. Access the material online at: https://dx.doi.org/10.17582/journal.pjz/20190202100216
Statement of conflict of interest
The authors have declared no conflict of interest.
REFERENCES
Ahmed, S., Sulaiman, S.A., Baig, A.A., Ibrahim, M., Liaqat S., Fatima, S., Jabeen, S., Shamim, N. and Othman N.H., 2018. Honey as a potential natural antioxidant medicine: An insight into its molecular mechanisms of action. Oxid. Med. Cell. Longevity, 8367846: 1-18. https://doi.org/10.1155/2018/8367846
Almasaudi, S.B., El-Shitany N.A., Abbas, A.T., Abdul-dayem, Ali, S.S., UA., Al-Jaouni, S.K. and Harakeh S., 2015. Antioxidant, anti-inflammatory, and antiulcer potential of manuka honey against gastric ulcer in rats. Oxid. Med. Cell. Longevity, 3643824. https://doi.org/10.1155/2016/3643824
Arai, K., Yamazaki, M. and Maeda, T., 2013. Influence of various treatments including povidone-iodine and healing stimulatory reagents in a rabbit ear wound model. Int. Wound J., 10: 542–548. https://doi.org/10.1111/j.1742-481X.2012.01016.x
Athar, M., Chaudhry, N.I. and Shakoor, 1996. Studies on end-to-end colonic anastomosis in the dog: A comparison of techniques. Acta Vet. Hung., 44: 349–356.
Benjamin, A.M. and Bowlin G.L., 2018. Honey-based templates in wound healing and tissue engineering. Bioengineering (Basel), 5: 46. https://doi.org/10.3390/bioengineering5020046
Bigliardi, P.L., Syed, A.L.A. and Hossam, Y.E.K., 2017. Povidone iodine in wound healing: A review of current concepts and practices. Int. J. Surg., 44: 260–268. https://doi.org/10.1016/j.ijsu.2017.06.073
Bindu, B., Ashish, B. and Girija, R., 2017. Temperature management under general anesthesia: Compulsion or option. J. Anaesthesiol. clin. Pharmacol., 33: 306–316. https://doi.org/10.4103/joacp.JOACP_334_16
Bobis, O., Daniel, S.D. and Adela, R.M., 2018. Honey and diabetes: the importance of natural simple sugars in diet for preventing and treating different type of diabetes. Oxid. Med. Cell. Longivity, pp. 1–12. https://doi.org/10.1155/2018/4757893
Chamanga, E.T., 2018. Clinical management of non-healing wounds. Nurs. Stand., 32: 48-63. https://doi.org/10.7748/ns.2018.e10829
Daniel, J.F., Emma, C.F. and Shannon, P.T., 2016. Cadexomer iodine provides superior efficacy against bacterial wound biofilms in vitro and in vivo. Wound Rep. Reg., 25: 31–24. https://doi.org/10.1111/wrr.12497
Eyarefe, D.O., David, I.K. and Theophilus, A.J., 2017. Enhanced electroscalpel incisional wound healing potential of honey in wistar rats. Int. J. Vet. Sci. Med., 5: 128–134. https://doi.org/10.1016/j.ijvsm.2017.10.002
Gulfraz M., Iftikhar, F.I. and Muhammad, A., 2011. Compositional analysis and antimicrobial activity of various honey types of Pakistan. Int. J. Fd. Sci. Tech., 46: 263–267. https://doi.org/10.1111/j.1365-2621.2010.02488.x
Hess, C.T., 2011. Checklist for factors affecting wound healing. Adv. Skin Wound Care, 24: 192. https://doi.org/10.1097/01.ASW.0000396300.04173.ec
Horng, H.C., Wen-Hsun, C. and Chang-Ching, Y., 2017. Estrogen effects on wound healing. Int. J. Mol. Sci., 7: 1–14.
Jeklova, E., Leva, L., Jaglic, Z. and Faldyna, M., 2008. Dexamethasone-induced immunosuppression: A rabbit model. Vet. Immunol. Immunopathol., 122: 231-240. https://doi.org/10.1016/j.vetimm.2007.11.011
Johani, M.M.K., Jensen, S.O. and Gosbell, I.B., 2017. Effect of cadexomer iodine on the microbial load and diversity of chronic non-healing diabetic foot ulcers complicated by biofilm in vivo. J. Antimicrob. Chemother., 72: 2093–2101. https://doi.org/10.1093/jac/dkx099
Kornblatt, A.P., Nicoletti, V.G. and Travaglia, A., 2016.The neglected role of copper ions in wound healing. J. Inorg. Biochem., 161: 1–8. https://doi.org/10.1016/j.jinorgbio.2016.02.012
Levine, J.M., 2017. The effect of oral medication on wound healing. Adv. Skin Wound Care, 30: 137–142. https://doi.org/10.1097/01.ASW.0000512112.60254.28
Lin, P.H., Matthew, S. and Haichang, L., 2018. Zinc in wound healing modulation. Nutrients, 10: 1–20. https://doi.org/10.3390/nu10010016
Lipman, N.S., Marini, R.P. and Erdman, S.E., 1990. A comparison of ketamine/xylazine and ketamine/xylazine/acepromazine anesthesia in the rabbit. Lab. Anim. Sci., 40: 395-398.
Marreiro, D.N. and Kyria, J.C.C., 2017. Zinc and oxidative stress. Curr. Mech. Antioxid., 6: 1–9. https://doi.org/10.3390/antiox6020024
Molan, P.C., 2011. The evidence and the rationale for the use of honey as wound dressing. Wound Prac. Res., 19: 204–220.
Molan and Rhodes., 2015. Honey: A biologic wound dressing. Wounds, 27: 141–151.
Samarghandian, S., Farkhondeh, T. and Samini, F., 2017. Honey and health: A review of recent clinical research. Pharmacol. Res., 9: 121–127.
Shi, C.M., Nakao, H. and Yamazaki, M., 2007. Mixture of sugar and povidone-iodine stimulates healing of MRSA-infected skin ulcers on db/db mice. Arch. Dermatol. Res., 299: 449. https://doi.org/10.1007/s00403-007-0776-3
Swaim, S.F., Bradley, D.M. and Hinkle, S.H., 2001. Wound contraction: Basic and clinical factors. Comp. Cont. Educ. Pract. Vet. North Am. Ed., 23: 20–35.
Tan, M.K., Adli, H., Tumiran, M.A., Abdulla, M.A. and Yusuff, K.M., 2012. The efficacy of gelam honey dressing towards excisional wound healing. Evid. Based Compllement. Alternat. Med., ID 805932: 1–6. https://doi.org/10.1155/2012/805932
To share on other social networks, click on any share button. What are these?










